02 July 2021: Review Paper
Clinical Rationale for a Routine Testing Schedule Using Donor-Derived Cell-Free DNA After Kidney Transplantation
Akshta Pai1ADEF*, Joshua T. SwanDOI: 10.12659/AOT.932249
Ann Transplant 2021; 26:e932249
Abstract
ABSTRACT: Kidney transplant recipients require meticulous clinical and laboratory surveillance to monitor allograft health. Conventional biomarkers, including serum creatinine and proteinuria, are lagging indicators of allograft injury, often rising only after significant and potentially irreversible damage has occurred. Immunosuppressive medication levels can be followed, but their utility is largely limited to guiding dosing changes or assessing adherence. Kidney biopsy, the criterion standard for the diagnosis and characterization of injury, is invasive and thus poorly suited for frequent surveillance. Donor-derived cell-free DNA (dd-cfDNA) is a sensitive, noninvasive, leading indicator of allograft injury, which offers the opportunity for expedited intervention and can improve long-term allograft outcomes. This article describes the clinical rationale for a routine testing schedule utilizing dd-cfDNA surveillance at months 1, 2, 3, 4, 6, 9, and 12 during the first year following kidney transplantation and quarterly thereafter. These time points coincide with major immunologic transition points after transplantation and provide clinicians with molecular information to help inform decision making.
Keywords: Cell-Free Nucleic Acids, Graft Rejection, Immunologic Surveillance, Kidney Transplantation, Pathology, Molecular, Tissue Donors, transplant recipients
Background
The United States kidney transplant wait list currently includes more than 90 000 candidates, with nearly 3000 more added every month. In 2019, only 23 401 kidney transplants were performed, exposing the dramatic gap between organ demand and availability [Organ Procurement and Transplantation Network, September 2020]. Many patients will either die before getting a transplant or become too sick to remain listed. Furthermore, despite improvements in median allograft survival over the last decade, over 10% of donor organs will go to repeat transplant recipients every year. A variety of strategies have been proposed to help address this imbalance, including better donor-recipient matching and increased utilization of marginal organs. The emergence of novel biomarkers such as donor-derived cell-free DNA (dd-cfDNA) may further aid in these efforts by allowing earlier identification and enhanced characterization of allograft injury.
Beyond the complications seen in the immediate postoperative period, attention turns to the multitude of threats to the long-term survival of both patient and allograft, including drug toxicity, opportunistic infections, recurrence of primary disease, and acute rejection. Current strategies for monitoring allograft health in the posttransplant period are limited, with conventional biomarkers such as creatinine and proteinuria serving as either lagging or nonspecific indicators of injury. Allograft tissue biopsy remains an integral part of posttransplant care, often being performed in response to clinical or laboratory changes suggestive of allograft dysfunction or as part of a routine surveillance protocol. However, this invasive, resource-intensive procedure, although considered a criterion standard for the diagnosis of rejection, carries risks of complications and is subject to the variability of histologic interpretation [1]. Measurement of dd-cfDNA, which has a half-life of only 30 to 60 min and is derived exclusively from allograft cells, has the potential to dramatically expand the diagnostic arsenal of the transplant nephrologist (Figure 1). Within transplantation, a growing body of evidence has shown the potential of the AlloSure dd-cfDNA test (CareDx, Brisbane, CA) to serve as an early indicator of allograft injury and a reliable biomarker to rule out rejection [2].
To date, there has been a single prospective multi-center validation study and 2 large ongoing registries evaluating the performance of AlloSure in posttransplant surveillance. The Diagnose Acute Rejection after kidney Transplant (DART) study (ClinicalTrials.gov identifier: NCT02424227) enrolled 384 renal transplant patients between April 2015 and May 2016 from 14 centers, either at 1 to 3 months after transplant or at the time of biopsy [2]. The DART study validated the performance of the AlloSure test as a dynamic and accurate marker of allograft injury. The demographics and clinical characteristics of the study cohort closely mirrored the United States transplant population, suggesting broad applicability of these findings [2]. Two ongoing studies include the Kidney Allograft Outcomes AlloSure Registry (KOAR) and the Outcomes of KidneyCare on Renal Allografts (OKRA) (ClinicalTrials.gov identifier: NCT03326076) registries, together enrolling a total of 4000 patients to evaluate the safety and efficacy outcomes in renal transplant recipients undergoing posttransplant surveillance with either AlloSure or KidneyCare. The latter is a multi-modality platform that includes AlloSure with AlloMap Kidney, a peripheral gene expression profiling assay currently in development, and iBox, a machine learning risk-stratification algorithm developed by the Paris Transplant Group [3]. All 3 studies (DART, KOAR, and OKRA) measure dd-cfDNA in accordance with the AlloSure Routine Testing Schedule (ARTS) at 1, 2, 3, 4, 6, 9, and 12 months in the first year after transplantation and then quarterly thereafter (Figure 2). The objective of this manuscript is to provide the clinical rationale and evidence for these time points, which were selected to coincide with major immunologic transition points during the first year after transplantation and subsequently with the timing of other typically ordered surveillance testing.
Immunosuppression in the First Year After Transplantation
Almost all patients undergoing kidney transplantation will receive induction therapy and maintenance immunosuppression. Induction therapy administered prior to or during transplantation involves either a T-cell depleting agent (rATG/Thymoglobulin or alemtuzumab) or interleukin (IL)-2 receptor blockade (basiliximab), usually in combination with high-dose steroids. Maintenance immunosuppression can vary considerably based on center preference and a patient’s individual risk for rejection. Commonly, a combination of 3 drugs is used, including a calcineurin inhibitor (CNI), an antimetabolite agent (mycophenolic acid or azathioprine), and steroids. For patients at low immunologic risk, co-stimulation blockade with belatacept is also considered as an alternative to CNI. A variety of other CNI-sparing and steroid-sparing strategies exist for patients at increased risk for CNI-related adverse events or those at low immunologic risk [4].
The first year after transplantation represents a series of major immunologic transition points, driven partly by the gradual reduction in the intensity of immunosuppression. In the immediate posttransplant period, steroids are tapered utilizing a variety of center-specific protocols, either to a low maintenance dose or off entirely. The goal trough levels for CNIs are adjusted downward after the first 8 to 12 weeks after transplantation. Finally, when T-cell-depleting induction therapies are used, their immunosuppressive effect begins to wane at 4 to 6 months [5]. The incidence of viral reactivation, including polyomavirus BK and cytomegalovirus as well as other opportunistic infections, is also highest immediately after transplantation, when the immune system is most suppressed. Addressing infectious complications can require further reduction of immunosuppression, necessitating a careful balancing act between enabling the host immune response and avoiding allograft rejection. The latter is a particularly important consideration during this time, with 7% to 8% of all recipients experiencing an episode of acute rejection during the first year after kidney transplantation according to the most recent Organ Procurement and Transplantation Network data [6]. These challenges are compounded by the limitations of conventional surveillance strategies and highlight the need for dynamic, noninvasive biomarkers that could inform clinical decision making during this critical period and beyond. The proposed dd-cfDNA surveillance protocol aims to account for both the complexity of posttransplant surveillance and the variation in clinical practice across transplant centers in the United States.
Rationale of dd-cfDNA Surveillance Time Points
MONTH 1:
The immediate posttransplant period is a proinflammatory state characterized by healing after surgical intervention, early immune recognition of the allograft, the presence of a foreign body (ureteral stent), and the potential for a broad array of early postoperative medical and surgical complications. Although dd-cfDNA levels can be measured as early as 2 weeks after transplantation, waiting for the 1-month mark can be preferable to allow for ureteral stent removal (which typically occurs anywhere from 2 to 6 weeks after transplantation) as well as recognition and resolution of common complications, including delayed graft function, impaired surgical wound healing, anastomotic leaks, urinomas, hematomas, ureteral obstruction, hydronephrosis, and lymphoceles [7,8]. Several ongoing studies evaluating dd-cfDNA levels in the context of these complications are ongoing and may offer additional insight on the interpretation and utility of the assay during this early period [9].
At the 1-month mark, obtaining the first dd-cfDNA level offers an opportunity to both establish an early baseline level and identify subclinical molecular injury. Significant elevations in dd-cfDNA levels at this juncture can signify evolving acute rejection, ongoing injury due to unidentified/uncorrected surgical complications, or early recurrence of primary disease.
MONTH 2:
By the end of the second month, several changes to maintenance immunosuppression regimens have usually occurred. Most programs utilizing steroids will have tapered them to a maintenance dose by 4 weeks after transplantation, and most patients on CNIs will have been stabilized on the doses required to achieve the target levels in this early period [10]. Obtaining a repeat dd-cfDNA level at this time point is thus useful to validate the previously established baseline, evaluate for subtle changes in molecular injury patterns after adjustments to maintenance immunosuppression, and potentially identify early rejection. Acute rejection is common in the first year after transplantation and is associated with recurrent rejection, alloimmunization, and adverse long-term outcomes [11,12]. The performance of dd-cfDNA in this respect far exceeds that of serum creatinine, making it an exceptionally helpful marker for early identification and intervention in these patients [2].
MONTH 3:
At the 3-month mark, programs that use CNIs as part of their maintenance regimens may decrease goal drug concentration levels, reducing the overall intensity of immunosuppression. An analysis of the DeKAF study showed this reduction was associated with an increased risk of acute rejection in the subsequent 3 months, highlighting the potential utility of dd-cfDNA surveillance in patients crossing this posttransplant time point [13]. For the minority of programs that perform protocol biopsies at the 3-month mark, dd-cfDNA levels could provide adjunctive information, helping risk stratify patients with borderline or nonspecific histologic changes [14]. For those programs not routinely performing protocol biopsies, dd-cfDNA levels at this time point would be even more valuable, identifying those patients with ongoing molecular injury during this critical period of immunosuppressive de-escalation.
MONTH 4:
The total dose of rATG or alemtuzumab administered as part of pretransplant induction is highly heterogeneous. Depending on the dose given, the effects of this induction can last anywhere between 4 to 6 months [5]. Given the high cost of rATG, it is not surprising that transplant centers have gradually reduced the total dose over time (from 10 mg/kg to 3 mg/kg in low-risk patients), hoping to minimize cost without affecting rejection rates. However, as patients transition from the protective immunosuppression offered by induction therapy to the coverage offered by oral medications alone, the risk of rejection rises, making this an important time point for assessment of molecular injury using dd-cfDNA, now easily comparable to well-established baseline levels over the preceding 3 months.
MONTH 6 AND MONTH 9:
Antibody-mediated rejection (AMR) is increasingly recognized as the leading cause of late transplant failure [15]. De novo donor-specific antibodies (dnDSA) have an established relationship with both acute and chronic AMR. Protocol-based surveillance for dnDSA is usually performed at least once between 3 and 12 months after transplantation, commonly at 6 and 9 months; testing can also be indicated following changes in immunosuppression, when there is concern for non-adherence, or in evaluating allograft dysfunction [16].
Inclusion of dd-cfDNA testing at these time points has significant value for several reasons. Testing at the 6-month mark allows reassessment of the baseline dd-cfDNA levels following the loss of the immunosuppressive effect provided by induction therapy. In addition, recent data has highlighted the relationship between dd-cfDNA levels and dnDSA. In a paper published in 2018, it was demonstrated that among DSA-positive transplant recipients, the combination of positive DSAs and an AlloSure level >1% had an 81% positive predictive value for AMR, rising to 89% when AlloSure levels exceeded 2.9% [17]. In a retrospective analysis of the DART cohort data presented at the American Transplant Congress, the same group also demonstrated that elevated AlloSure levels were an independent risk factor for dnDSA formation [18]. The associations between inflammation and alloimmunization are well known; however, these data highlight the potential of dd-cfDNA as a sensitive biomarker to characterize these processes on a molecular level, identifying patients at risk of developing dnDSA or those with subclinical allograft injury in the context of existing DSAs.
MONTH 12:
The end of the first posttransplant year is accompanied by clinical evaluation and another round of extensive laboratory testing. For those centers not performing surveillance biopsies at the 1-year mark, the addition of dd-cfDNA testing helps provide reassurance that less sensitive parameters like serum creatinine and urine proteinuria assessment are not overlooking subclinical molecular injury or early evolving rejection. For those centers that do include histologic assessment at this stage, dd-cfDNA levels can help stratify and provide insight as to the long-term clinical significance of the borderline or subclinical histological changes that can be identified [14]. Eventually, dd-cfDNA data collected during the first posttransplant year could provide further risk stratification, helping predict long-term outcomes in kidney transplant recipients together with other metrics currently utilized for this purpose [19].
Quarterly Surveillance: Year 2 and Beyond
Most posttransplant patients continue to have blood work at 3-month intervals after the first year, allowing clinicians to detect changes in allograft function, monitor levels of immunosuppressive agents, identify drug toxicities, and detect rejection. The addition of dd-cfDNA to this regimen empowers clinicians by providing them with an accurate, sensitive, and noninvasive biomarker of allograft injury, facilitating earlier identification of evolving rejection when scores are rising and the relative certainty of immune quiescence when dd-cfDNA scores are low. In addition, we believe evidence is building for other applications of dd-cfDNA levels in the longitudinal management of transplant recipients.
Among these is a retrospective analysis of the DART cohort which demonstrated that an elevated dd-cfDNA level (≥1.0%) during the first year after transplantation was associated with a significant decline in eGFR (median change of −25%) and an eGFR <30 mL/min in the second year [20]. This association is particularly important as declines in eGFR have been shown to correlate well with hard outcomes in kidney transplantation and are often utilized as surrogate endpoints in clinical trials [21]. Taken together with previously described evidence linking dd-cfDNA levels and dnDSA formation, the role of molecular injury and inflammation in driving adverse transplant outcomes becomes increasingly clear, as does the considerable potential for dd-cfDNA-guided risk stratification and longitudinal surveillance of transplant recipients. It is not difficult to envision dd-cfDNA levels one day being utilized to tailor immunosuppressive regimens much in the same way we use trough levels to adjust CNI dosing today, a strategy that could reduce the incidence of adverse effects associated with immunosuppression. Larger, well-designed studies exploring the utility of dd-cfDNA in these settings are needed, but emerging evidence is very encouraging.
Conclusions
The rationale for routine dd-cfDNA surveillance is grounded in the utility of evaluating molecular injury patterns at key immunologic transition points following kidney transplantation. The frequency and timing of testing accounts for the increased risk of rejection in the early posttransplant period and the added value of molecular information, when combined with other routine testing throughout the life of the allograft. Ongoing large-scale studies utilizing the proposed regimen are expected to provide additional insight into the impact of dd-cfDNA surveillance on important clinical outcomes among kidney transplant recipients.
Figures
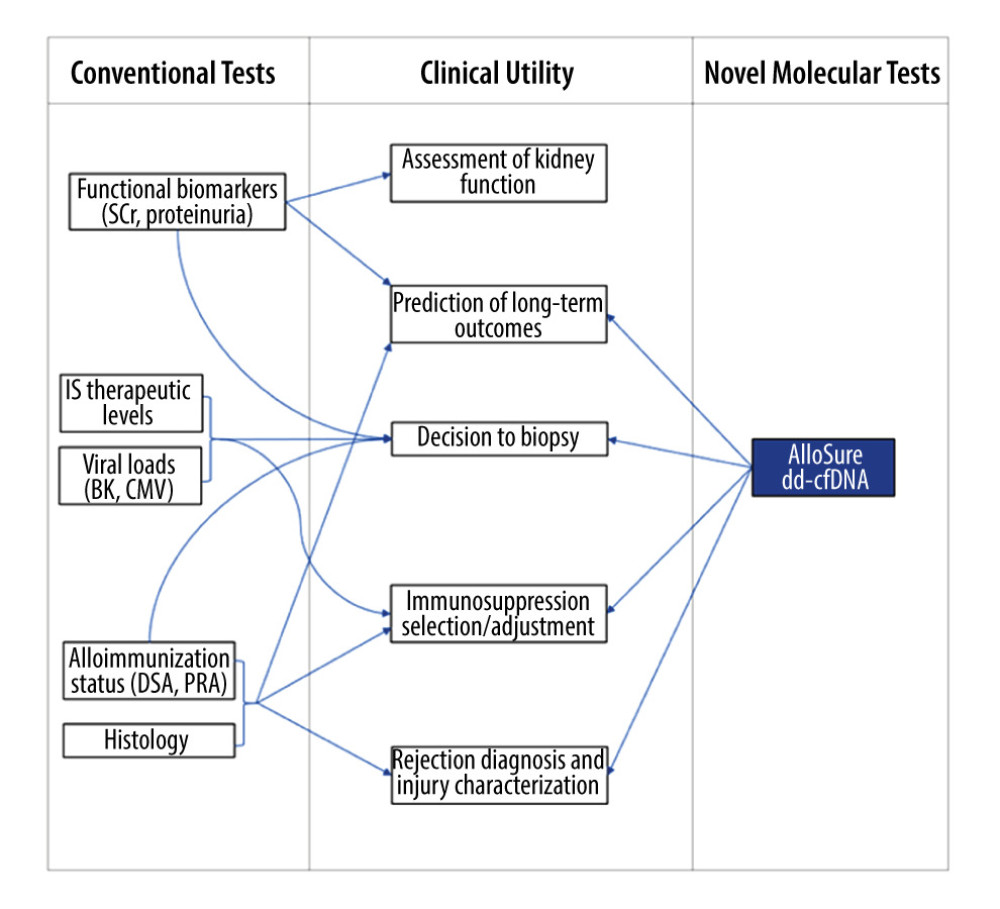 Figure 1. Graphic depicting the potential added value of donor-derived cell-free DNA (dd-cfDNA) surveillance for augmenting clinical decision making in posttransplant management. SCr – serum creatinine; DSA – donor-specific antibodies; PRA – panel reactive antibodies; IS – immunosuppressive therapy.
Figure 1. Graphic depicting the potential added value of donor-derived cell-free DNA (dd-cfDNA) surveillance for augmenting clinical decision making in posttransplant management. SCr – serum creatinine; DSA – donor-specific antibodies; PRA – panel reactive antibodies; IS – immunosuppressive therapy. 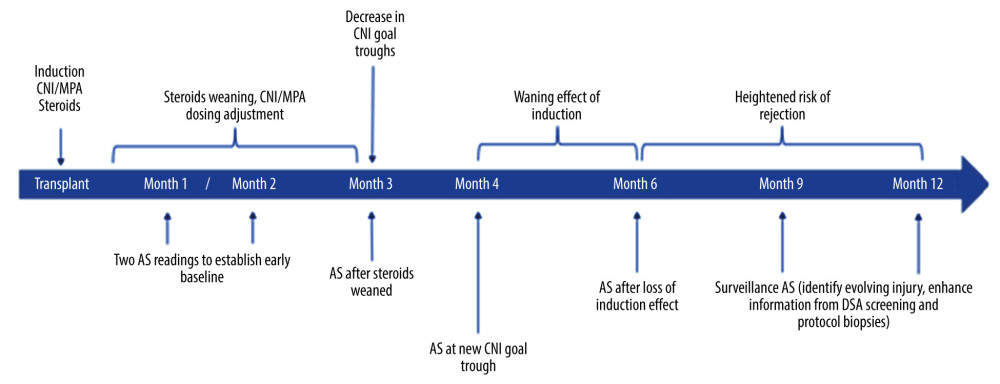 Figure 2. Time points and rationale for donor-derived cell-free DNA (dd-cfDNA) testing for the proposed AlloSure Routine Testing Schedule (ARTS). AS – AlloSure; CNI – calcineurin inhibitor; MPA – mycophenolic acid; DSA – donor-specific antibodies.
Figure 2. Time points and rationale for donor-derived cell-free DNA (dd-cfDNA) testing for the proposed AlloSure Routine Testing Schedule (ARTS). AS – AlloSure; CNI – calcineurin inhibitor; MPA – mycophenolic acid; DSA – donor-specific antibodies. References
1. Knight SR, Thorne A, Lo Faro ML, Donor-specific cell-free DNA as a biomarker in solid organ transplantation. A systematic review: Transplantation, 2019; 103(2); 273-83
2. Bloom RD, Bromberg JS, Poggio ED, Cell-free DNA and active rejection in kidney allografts: J Am Soc Nephrol, 2017; 28(7); 2221-32
3. Loupy A, Aubert O, Orandi BJ, Prediction system for risk of allograft loss in patients receiving kidney transplants: International derivation and validation study: BMJ, 2019; 366; l4923
4. Lim MA, Kohli J, Bloom RD, Immunosuppression for kidney transplantation: Where are we now and where are we going?: Transplant Rev (Orlando), 2017; 31(1); 10-17
5. Gurkan S, Luan Y, Dhillon N, Immune reconstitution following rabbit antithymocyte globulin: Am J Transplant, 2010; 10(9); 2132-41
6. Hart A, Smith JM, Skeans MA, OPTN/SRTR 2018 annual data report: Kidney: Am J Transplant, 2020; 20(S1); 20-130
7. Levine MA, Schuler T, Gourishankar S, Complications in the 90-day postoperative period following kidney transplant and the relationship of the Charlson Comorbidity Index: Can Urol Assoc J, 2017; 11(12); 388-93
8. Siedlecki A, Irish W, Brennan DC, Delayed graft function in the kidney transplant: Am J Transplant, 2011; 11(11); 2279-96
9. Gielis EM, Beirnaert C, Dendooven A, Plasma donor-derived cell-free DNA kinetics after kidney transplantation using a single tube multiplex PCR assay: PLoS One, 2018; 13(12); e0208207
10. Pallardó LM, Oppenheimer F, Guirado L, Calcineurin inhibitor reduction based on maintenance immunosuppression with mycophenolate mofetil in renal transplant patients: POP study: Transplant Proc, 2007; 39(7); 2187-89
11. Opelz G, Dohler BCollaborative Transplant Study Report, Influence of time of rejection on long-term graft survival in renal transplantation: Transplantation, 2008; 85(5); 661-66
12. El Ters M, Grande JP, Keddis MT, Kidney allograft survival after acute rejection, the value of follow-up biopsies: Am J Transplant, 2013; 13(9); 2334-41
13. Israni AK, Riad SM, Leduc R, Tacrolimus trough levels after month 3 as a predictor of acute rejection following kidney transplantation: A lesson learned from DeKAF Genomics: Transpl Int, 2013; 26(10); 982-89
14. Stites E, Kumar D, Olaitan O, High levels of dd-cfDNA identify patients with TCMR 1A and borderline allograft rejection at elevated risk of graft injury: Am J Transplant, 2020; 20(9); 2491-98
15. Sellares J, de Freitas DG, Mengel M, Understanding the causes of kidney transplant failure: The dominant role of antibody-mediated rejection and nonadherence: Am J Transplant, 2012; 12(2); 388-99
16. Tait BD, Susal C, Gebel HM, Consensus guidelines on the testing and clinical management issues associated with HLA and non-HLA antibodies in transplantation: Transplantation, 2013; 95(1); 19-47
17. Jordan SC, Bunnapradist S, Bromberg JS, Donor-derived cell-free DNA identifies antibody-mediated rejection in donor specific antibody positive kidney transplant recipients: Transplant Direct, 2018; 4(9); e379
18. Jordan SC, Bunnapradist S, Bromberg JS, Donor-derived cell-free DNA identifies antibody-mediated rejection in donor specific antibody positive kidney transplant recipients: Transplant Direct, 2018; 4(9); e379
19. Menon MC, Murphy B, Heeger PS, Moving biomarkers toward clinical implementation in kidney transplantation: J Am Soc Nephrol, 2017; 28(3); 735
20. Alhamad T, Poggio ED, Hiller D, The Use of dd-cfDNA as a predictive tool for outcome decreased kidney function [abstract]: Am J Transplant, 2019; 19 Abstract number: 206
21. Clayton PA, Lim WH, Wong G, Chadban SJ, Relationship between eGFR decline and hard outcomes after kidney transplants: J Am Soc Nephrol, 2016; 27(11); 3440-46
Figures
 Figure 1. Graphic depicting the potential added value of donor-derived cell-free DNA (dd-cfDNA) surveillance for augmenting clinical decision making in posttransplant management. SCr – serum creatinine; DSA – donor-specific antibodies; PRA – panel reactive antibodies; IS – immunosuppressive therapy.
Figure 1. Graphic depicting the potential added value of donor-derived cell-free DNA (dd-cfDNA) surveillance for augmenting clinical decision making in posttransplant management. SCr – serum creatinine; DSA – donor-specific antibodies; PRA – panel reactive antibodies; IS – immunosuppressive therapy. Figure 2. Time points and rationale for donor-derived cell-free DNA (dd-cfDNA) testing for the proposed AlloSure Routine Testing Schedule (ARTS). AS – AlloSure; CNI – calcineurin inhibitor; MPA – mycophenolic acid; DSA – donor-specific antibodies.
Figure 2. Time points and rationale for donor-derived cell-free DNA (dd-cfDNA) testing for the proposed AlloSure Routine Testing Schedule (ARTS). AS – AlloSure; CNI – calcineurin inhibitor; MPA – mycophenolic acid; DSA – donor-specific antibodies. In Press
15 Mar 2024 : Review article
Approaches and Challenges in the Current Management of Cytomegalovirus in Transplant Recipients: Highlighti...Ann Transplant In Press; DOI: 10.12659/AOT.941185
18 Mar 2024 : Original article
Does Antibiotic Use Increase the Risk of Post-Transplantation Diabetes Mellitus? A Retrospective Study of R...Ann Transplant In Press; DOI: 10.12659/AOT.943282
20 Mar 2024 : Original article
Transplant Nephrectomy: A Comparative Study of Timing and Techniques in a Single InstitutionAnn Transplant In Press; DOI: 10.12659/AOT.942252
28 Mar 2024 : Original article
Association Between FEV₁ Decline Rate and Mortality in Long-Term Follow-Up of a 21-Patient Pilot Clinical T...Ann Transplant In Press; DOI: 10.12659/AOT.942823
Most Viewed Current Articles
05 Apr 2022 : Original article
Impact of Statins on Hepatocellular Carcinoma Recurrence After Living-Donor Liver TransplantationDOI :10.12659/AOT.935604
Ann Transplant 2022; 27:e935604
12 Jan 2022 : Original article
Risk Factors for Developing BK Virus-Associated Nephropathy: A Single-Center Retrospective Cohort Study of ...DOI :10.12659/AOT.934738
Ann Transplant 2022; 27:e934738
22 Nov 2022 : Original article
Long-Term Effects of Everolimus-Facilitated Tacrolimus Reduction in Living-Donor Liver Transplant Recipient...DOI :10.12659/AOT.937988
Ann Transplant 2022; 27:e937988
15 Mar 2022 : Case report
Combined Liver, Pancreas-Duodenum, and Kidney Transplantation for Patients with Hepatitis B Cirrhosis, Urem...DOI :10.12659/AOT.935860
Ann Transplant 2022; 27:e935860








