18 February 2022: Original Paper
Impact on Waitlist Outcomes from Changes in the Medical Eligibility of Candidates for Simultaneous Liver-Kidney Transplantation Following Implementation of the 2017 Organ Procurement and Transplantation Network/United Network for Organ Sharing Policy in the United States
Shingo Shimada1ADEF*, Toshihiro Kitajima1BCE, Yukiko Suzuki1BD, Yasutaka Kuno1BD, Tayseer Shamaa1BC, Tommy IvanicsDOI: 10.12659/AOT.934850
Ann Transplant 2022; 27:e934850
Abstract
BACKGROUND: The new simultaneous liver-kidney transplantation (SLK) listing criteria in the United States was implemented in 2017. We aimed to investigate the impact on waitlist and post-transplantation outcomes from changes in the medical eligibility of candidates for SLK after policy implementation in the United States.
MATERIAL AND METHODS: We analyzed adult primary SLK candidates between January 2015 and March 2019 using the Organ Procurement and Transplant Network/United Network for Organ Sharing (OPTN/UNOS) registry. We compared waitlist practice, post-transplantation outcomes, and final transplant graft type in SLK candidates before and after the policy.
RESULTS: A total of 4641 patients were eligible, with 2975 and 1666 registered before and after the 2017 policy, respectively. The daily number of SLK candidates was lower after the 2017 policy (3.25 vs 2.89, P=0.01); 1956 received SLK and 95 received liver transplant alone (LTA). The proportion of patients who eventually received LTA was higher after the 2017 policy (7.9% vs 3.0%; P<0.001). The 1-year graft survival rate was worse in patients with LTA than in those with SLK (80.5% vs 90.4%; P=0.003). The adjusted risk of 1-year graft failure in patients with LTA was 2.01 (95% confidence interval 1.13-3.58, P=0.01) compared with patients with SLK among the SLK candidates.
CONCLUSIONS: Although the number of registrations for SLK increased, the number of SLK transplants decreased, and the number of liver transplants increased. LTA in this patient cohort was associated with worse post-transplantation outcomes.
Keywords: Kidney Transplantation, Liver, Patient Outcome Assessment, Waiting Lists, Adult, Humans, Liver Transplantation, policy, Risk Factors, Tissue and Organ Procurement, United States
Background
Simultaneous liver-kidney transplantation (SLK) is indicated for patients with end-stage liver disease and severe renal impairment [1,2]. In the United States, the proportion of SLK candidates has increased from 2.0% in 2005 to 6.6% in 2015. Similarly, the actual number of patients who undergo SLK has increased from 5.6% in 2005 to 9.4% in 2015 [3].
The prevailing organ shortage remains a challenge in the fields of both liver and kidney transplantation. Given the increase in the number of patients who require SLK [4], creating equitable allocation rules for SLK is crucial. Within this context, despite general allocation guidelines by the Organ Procurement and Transplantation Network/United Network for Organ Sharing (OPTN/UNOS), the indications for SLK between centers often varied. In 2017, the new SLK policy was implemented and resulted in homogenizing SLK listing criteria nationally [5,6]. The eligibility criteria for SLK in these guidelines include (1) chronic kidney disease, with an estimated glomerular filtration rate (eGFR) ≤60 for greater than 90 consecutive days before listing or ≤30 at listing, or end-stage renal disease on maintenance dialysis; (2) sustained acute kidney injury requiring acute dialysis for 6 weeks or longer, an eGFR ≤25 for 6 weeks or more, or dialysis and eGFR ≤25 for 6 consecutive weeks; and (3) metabolic disease. To be eligible for SLK, candidates must meet at least 1 of these criteria [7]. In addition, SLK candidates must fulfill at least 1 of the criteria throughout the entire duration of the transplant listing [7]. If a candidate no longer meets the criteria, they no longer qualify for SLK and instead become listed for a liver transplant alone (LTA). As the kidney function in liver transplantation candidates can fluctuate during the waiting time, it is possible that some patients who are initially listed for SLK have an improvement in their kidney function and no longer meet the SLK requirements. In this case, they would undergo LTA. Several studies have evaluated the impact of the implementation of SLK policy in the United States [8,9]. Cullaro et al reported the possible impact of SLK policy on post-transplant outcomes using the OPTN/UNOS dataset before the policy implementation and concluded that the policy implementation might not improve survival after transplantation [8]. Recently, Wilk et al reported that the number of kidney transplantation after LTA and the transplant rate in patients listed for kidney transplantation after LTA increased, and post-transplant outcomes in patients who received SLK after the policy implementation were similar to those before the implementation. They did not compare the outcomes according to final graft types (SLK or LTA) in patients who were initially listed for SLK [9].
We aimed to investigate the impact of changes in the medical eligibility of SLK candidates in the United States according to the OPTN/UNOS SLK policy on waitlist outcome, post-transplant outcome, and transplant graft type (SLK or LTA) in SLK listings from the OPTN/UNOS transplant registry.
Material and Methods
STUDY POPULATION:
This study was approved for an Institutional Review Board waiver after review.
We analyzed data from the OPTN/UNOS Standard Transplant and Research files for liver transplants between January 1, 2015, and March 31, 2019, for information from patients listed for primary SLK in the United States. The study period was set from January 1, 2015, to March 31, 2019. Exclusion criteria were patients younger than 18 years old, patients initially listed for either LTA, retransplant, or combined organ transplant with the thoracic organs, intestines, and/or pancreas. The study period was divided into 2 eras: before the 2017 policy, which included patients listed on the SLK list from January 1, 2015, to July 31, 2017, and after the 2017 policy, which included patients listed for SLK from September 1, 2017, to March 31, 2019. Because the new OPTN/UNOS SLK policy was implemented in August 2017, patients listed in August 2017 were excluded as a 1-month washout period to minimize the effect on the waitlist practice by the 2 different policies. There were 201 patients with dual listings, of which 33 died on the waitlist and 111 underwent transplantation and were among the listed patients eligible for this study. To avoid double-counting listed patients, those who had either death or transplant at the time of waitlist removal were eligible for the study, and patients who were improved or censored were excluded from the study.
COVARIATES:
Continuous variables included age, body mass index (BMI), model for end-stage liver disease (MELD) score, waiting time, eGFR, serum creatinine, serum total bilirubin, prothrombin time-international normalized ratio (PT-INR), serum sodium, and cold ischemia time. Sex, moderate/severe ascites, dialysis requirement, grade 3/4 encephalopathy, need for mechanical ventilation, portal vein thrombosis, and donation after circulatory death (DCD) donor were considered as binary variables. Multilevel categorical variables included ethnicity (White, Black, Hispanic, and other), Karnofsky score (10–30%, 40–60%, and 70–100%), share type (local, regional, and national), recipient age (<50, 50–59, and 60 years or older), donor age (<40, 40–59, and 60 years or older), MELD (MELD-Na[sodium]) score (6–29, 30–34, and 35 or higher), and serum sodium value (<135, 135–144, and 145 mEq/L or higher).
SIMULTANEOUS LIVER-KIDNEY TRANSPLANT WAITLIST PRACTICE AND OUTCOMES:
We compared the numbers of newly listed patients for primary SLK between the 2 eras. Also, liver transplant waitlist outcomes, including waitlist mortality and transplant probability, were compared between the 2 eras by using a competing risk analysis, which we used for our previous study [10]. Competing risk events included improvement on the waitlist, deceased donor transplant, death, and extreme sickness. If none of the abovementioned events had occurred before the end of the set period, patients were censored. Patients who received living donor transplants were censored at the time of transplantation. Patients listed before the 2017 policy were censored on the last day of each era (July 31, 2017) in the waitlist outcome analysis to minimize the effects of different SLK listing criteria. Fine-gray proportional hazards were adjusted for recipient characteristics at listing, including recipient age, sex, race, MELD/MELD-Na score, serum sodium, presence of ascites, Karnofsky score, hepatic encephalopathy, and dialysis requirement. Liver transplant waitlist outcomes were analyzed and stratified by MELD score categories: lower MELD score group: score of 29 or lower; mid MELD score group: score of 30–34; and higher MELD score group: score of 35 or higher. The proportions of actual transplant graft type (SLK or LTA) in SLK listings were also compared between the 2 eras.
POST-TRANSPLANT OUTCOMES:
Among the patients initially listed for SLK, the risks of 1-year graft loss and post-transplantation mortality were compared between the transplant types (SLK vs LTA). Risks were adjusted for donor age, donor race, donor BMI, DCD donor, recipient race, and cold ischemia time and the following variables at the time of transplantation: recipient age, encephalopathy, ascites, mechanical ventilation, dialysis, recipient diabetes, recipient BMI, MELD score, serum sodium, Karnofsky score, and UNOS regions.
Thereafter, we compared 1-year post-transplant outcomes between the 2 eras in patients who received SLK. Patients who were listed before the 2017 policy and transplanted after the 2017 policy were excluded in the post-transplant outcome analyses to eliminate the effect of policy change on outcomes of patients before the 2017 policy. Risk factors for post-SLK mortality and graft loss were evaluated by adjusting risks for the following variables at the time of SLK: recipient age, recipient race, encephalopathy, ascites, mechanical ventilation, dialysis, recipient diabetes, recipient MELD score, Karnofsky score, BMI, serum sodium, cold ischemia time, donor age, donor race, donor BMI, DCD graft, and UNOS regions.
STATISTICAL ANALYSIS:
Data were represented using percentages for discrete variables and the median and interquartile range (IQR) for continuous variables. The Mann-Whitney U test was used for continuous variables, and the chi-squared test was used for discrete variables. For the waitlist analysis, we used a cumulative incidence approach to account for the presence of a competing risk of transplantation on waitlist dropout due to mortality [10]. The Gray test was used for comparison of waitlist mortality and transplantation probability rates [11]. Multivariable models were created to define factors that affected waitlist outcomes. Fine-Gray proportional hazard regression models were used for comparisons of waitlist mortality/dropout and transplant probability [12]. Post-transplant survival was evaluated using Kaplan-Meier curve analysis and compared by log-rank tests. A multivariable Cox regression model assessed hazards of post-transplant mortality. A P value <0.05 was considered statistically significant for all analyses. All statistical analyses were done using SPSS version 25 (IBM Corp, Armonk, NY, USA) and R version 3.5.1 (R Foundation for Statistical Computing, Vienna, Austria).
Results
SLK TRANSPLANT WAITLIST PATIENT CHARACTERISTICS:
Between January 1, 2015, and March 31, 2019, a total of 5320 patients were listed for SLK. Patients under 18 years of age (n=113), with combined transplant with thoracic, intestinal, and/or pancreatic (n=49), listed for retransplantation (n=469), and listed in August 2017 (n=106) were excluded. The total number of patients eligible for this study was 4641 (Figure 1). Among them, 2975 were listed before the 2017 policy and 1666 were listed after the policy. Table 1 shows the comparison of patient characteristics in the SLK listing between the eras. The daily number of SLK listings was significantly lower after the 2017 policy than before the policy (3.25/day in before vs 2.89/day in after, P=0.01). Patients’ eGFR was significantly lower after compared with before the 2017 policy.
SIMULTANEOUS LIVER-KIDNEY TRANSPLANT WAITLIST OUTCOMES:
Risks of 90-day SLK waitlist mortality were similar before and after the 2017 policy (adjusted hazard ratio [aHR] 0.83, 95% confidence interval [CI] 0.67–1.02, P=0.08 [ref. before the 2017 policy]) (Table 2). When analyzing waitlist outcomes stratified by MELD score groups, waitlist mortality in patients with a MELD score of 29 or lower was significantly lower after compared with before the 2017 policy (aHR 0.72, 95% CI 0.52–0.99, P=0.04). Transplantation probability was significantly higher after compared with before the 2017 policy (aHR 1.12, 95% CI 1.00–1.26, P=0.04) (Table 2). In patients with a MELD score of 35 or higher, transplantation probability was significantly higher after compared with before the 2017 policy (aHR 1.24, 95% CI 1.02–1.51, P=0.03).
TREND OF ACTUAL TRANSPLANT TYPE IN SIMULTANEOUS LIVER-KIDNEY TRANSPLANT LISTINGS:
Among the 4641 SLK patients listed, 95 patients received LTA. Of these, 40 and 55 were before and after the 2017 policy, respectively. The proportion of patients who underwent LTA in SLK listings was significantly higher after the 2017 policy (3.0% before vs 7.9% after; P<0.001) (Figure 2). When SLK listings were stratified by MELD score groups, the proportion of patients who underwent LTA was significantly higher after compared with before the 2017 policy across all MELD score categories (MELD score 29 or lower, 5.7% vs 2.5%, P=0.006; MELD score 30–34, 9.7% vs 2.5%, P=0.009; MELD score 35 or higher, 12.5% vs 4.7%, P=0.004). This difference was most pronounced in the high MELD score category.
Among the patients who underwent LTA, 5 underwent kidney transplantation alone after LTA. Although there was no significant difference between the 2 eras, the proportion of patients who underwent kidney transplantation separately after LTA increased after the 2017 policy (2.5% [1/40] before vs 7.3% [4/55] after,
POST-TRANSPLANT OUTCOMES BETWEEN SIMULTANEOUS LIVER-KIDNEY TRANSPLANTATION AND LIVER TRANSPLANTATION ALONE:
Of the 4641 SLK patients listed, 1956 underwent SLK and 95 underwent LTA. The comparisons of characteristics of patients who underwent SLK and LTA are shown in Table 3. Although kidney function was better in patients who received LTA, median serum total bilirubin and PT-INR were significantly higher in patients who received LTA than in those who received SLK. The proportion of patients with mechanical ventilation and higher MELD scores was significantly higher in patients who received LTA. Also, the waiting time was shorter and donor age was older in patients who received LTA than in those who underwent SLK.
The 1-year graft survival rate in the patients who underwent LTA who were initially listed for SLK was significantly lower than in patients who underwent SLK (80.5% vs 90.4%, P=0.003) (Figure 3). After adjusting risks of 1-year graft loss and mortality for recipient and donor characteristics, the adjusted risks of 1-year graft loss and mortality in patients with LTA were significantly higher than in patients with SLK (graft loss: aHR 2.01, 95% CI 1.13–3.58, P=0.01; mortality: aHR 2.16, 95% CI 1.19–3.92, P=0.01) (Table 4).
COMPARISON OF SLK OUTCOMES BEFORE AND AFTER THE 2017 POLICY:
The characteristics of SLK recipients before and after the 2017 policy are shown in Table 5. Compared with before the policy implementation, the proportion of the patients with hyponatremia was significantly higher, eGFR and serum total bilirubin were lower, and cold ischemia time was shorter after implementation. On comparing SLK outcomes between the eras, the 1-year graft survival rate was significantly worse after the policy (86.3%) than before (91.4%) (P=0.01) (Figure 4). The multivariable Cox regression model showed that SLK after the 2017 policy was a risk factor for 1-year graft loss (aHR 1.57, 95% CI 1.11–2.22, P=0.01) along with mechanical ventilation (aHR 1.78, 95% CI 1.13–2.79, P=0.01), hyponatremia (<135 mEq/L: aHR 1.44, 95% CI 1.03–2.02, P=0.03), hypernatremia (≥145 mEq/L: aHR 2.70, 95% CI 1.19–6.11, P=0.01), and older donor age (40–59 years: aHR 1.69, 95% CI 1.20–2.38, P=0.002; ≥60 years: aHR 3.32, 95% CI 1.72–6.44, P<0.001) (Table 6). Infection was the most common cause of death in both eras (36.3% [37/102] before vs 32.6% [15/46] after the policy, P=0.09) within 1 year after SLK, although there was no significant difference between the 2 eras (Table 7).
COMPARISON OF LTA OUTCOMES BEFORE AND AFTER THE 2017 POLICY:
The proportion of the patients with moderate or severe ascites and portal vein thrombosis was lower (Table 8) after the policy than before. The median eGFR at LTA was better after the 2017 policy, although the difference was not statistically significant (33.8 mL/min vs 26.2 mL/min, P=0.33). Other covariates, such as kidney function, were comparable. On comparing LTA outcomes between eras, the 1-year graft survival rate was similar before (79.8%) and after the 2017 policy (81.3%) (P=0.89; Figure 5). Among patients who underwent LTA, infection was also the most common cause of death within 1 year after transplantation (57.1%; 8 of 14 died within 1 year after LTA; Table 9).
Discussion
We revealed that the number of SLK listings significantly decreased and the proportion of patients who received LTA from the SLK listings was significantly higher after the implementation of the 2017 policy. During the waiting time, patients can become disqualified for an SLK and instead undergo LTA. The post-transplant outcomes in LTA recipients were significantly worse than those of SLK recipients. These findings indicate that, while the new SLK policy might improve the regulation of SLK listings, the access to the final transplantation graft type needs to be carefully considered, especially in patients with marginal kidney function.
In this study, we evaluated the post-transplant outcomes in patients initially listed for SLK. Of 4641 patients listed for SLK, 95 and 1956 received LTA and SLK, respectively. Although recipients of LTA had significantly better kidney function than did SLK recipients at the time of transplantation, their kidney function remained marginal (median eGFR in the LTA group, 31.1 mL/min), predisposing them to an increased risk of post-transplant kidney failure. Kidney dysfunction is a well-known poor prognostic factor for morbidity and mortality after liver transplantation [1,13]. In line with this, the 1-year graft survival rate in the patients with LTA was significantly worse than that of patients with SLK, which might be related to their suboptimal post-transplant recovery of kidney function. Also, LTA recipients had a significantly higher MELD score and required mechanical ventilation more often at the time of transplantation, although LTA recipients had a significantly better kidney function than did SLK recipients. Median serum total bilirubin and PT-INR were significantly higher in patients who received LTA. Even patients who had more severe liver status among patients who were initially listed for SLK might receive LTA if they had improved kidney function. It is conceivable that they may have been unable to wait for 2 organs, and therefore a decision might have been made to proceed with LTA. Actually, the waiting time was shorter in patients who received LTA than in those with SLK. However, given the significantly poor risk-adjusted post-transplant outcomes observed, careful assessments are crucial when proceeding with LTA in patients with marginal kidney function, even if kidney function improves over that at the listing for SLK.
According to the simulation by Cullaro et al using the OPTN/UNOS dataset before the policy implementation, patients who met the new SLK criteria but underwent LTA in the past had significantly decreased post-transplantation mortality compared with SLK recipients [8]. Our study evaluated patients who were listed for SLK before and after the policy implementation. In our study, post-transplant outcomes of patients who underwent LTA but who initially listed for SLK were different from the results of the simulation. While studies have shown that the post-transplant outcomes of LTA recipients are significantly worse than those of SLK recipients [6,14], no reports have compared the post-transplant outcomes between SLK and LTA recipients who were initially listed for SLK. Recently, Samoylova et al reported that post-transplantation patient survival in patients who had dialysis requirements at listing or at transplantation or eGFR <25 trended toward a higher rate of SLK than of LTA before the policy change, and this was not different after the policy change [15]. Because our study focused on patients who were initially listed for SLK, our study is different from their study.
The proportion of patients listed for SLK who eventually received LTA was significantly higher after implementation of the SLK policy (7.9% after vs 3.0% before,
Worsened SLK outcomes after policy implementation was another important finding of the present study. According to the report by Wilk et al, post-transplant outcomes in SLK recipients were similar before and after policy implementation [9], which was different from our results; however, the patient cohort of our study was different from this cohort, and therefore, our results might be different from the findings by Wilk et al. There are possible factors that could have worsened the outcomes in SLK recipients. First, eGFR at transplantation was lower after than before the 2017 policy; therefore, patients might be sicker in the era after the policy implementation. Second, while the new SLK policy was introduced in 2017, the MELD-Na score-based allocation was introduced for liver allocation in January 2016 [16,17]. Therefore, patients after the 2017 policy may be more affected by the introduction of the MELD-Na score-based allocation. We observed that the proportion of patients with hyponatremia was significantly higher after the 2017 policy than before the policy. According to the analysis of risk factors for graft loss and mortality after SLK, hyponatremia was considered an independent risk factor along with older donor age and mechanical ventilation. Hyponatremia [18,19] and the need for mechanical ventilation [20] were previously reported as poor prognostic factors after liver transplantation. The findings of these reports were consistent with our results. The higher number of SLK recipients with hyponatremia might have led to worse post-transplantation outcomes after the 2017 policy.
This study revealed that transplant probability in SLK listings improved after the 2017 policy. This improvement might be due not only to policy implementation but to other reasons as well. First, the recent increase in the number of deceased donors might improve transplantation access [21]. Second, it was reported that the MELD-Na score-based allocation, which was implemented in January 2016, improved waitlist outcomes, including lowering mortality and increasing the probability of transplantation [17]. Patients after the 2017 policy might benefit from the MELD-Na-based allocation.
A critical feature in the new SLK policy is “safety net” kidney transplantation. The safety net prioritizes the kidney allocation for LTA recipients who develop kidney failure after their transplant [22]. We found that the proportion of patients who underwent subsequent kidney transplants after LTA was 2.5% (1/40) before the 2017 policy and 7.3% (4/55) after the policy. A meta-analysis based on 38 studies showed that the incidence rate of kidney failure requiring renal replacement therapy after LTA was 7.0% [23]. The risk of kidney failure in those LTA recipients might be similar to that reported in the meta-analysis. However, it should be noted that the most common cause of death in this population after transplantation was infection. There might be patients who are precluded from a kidney transplant owing to other morbidities. There is a possibility that a larger number of patients progressed to kidney failure after LTA. It is conceivable that an SLK for this population might have led to better post-transplant outcomes and saved medical and financial resources. Because the actual number of patients whose transplant graft type was switched from SLK to LTA was low, utilizing kidney grafts for this particular population was unlikely to affect the waitlist practice in kidney transplant-alone candidates.
This study had several limitations. This was a retrospective study using the OPTN/UNOS registry, which lacks detailed post-transplantation clinical data, such as the trend of liver and kidney graft function after transplantation. We were unable to evaluate the reasons for transplant graft change during the waiting time, and it was assumed that these patients might have had an improvement in kidney function, leading to disqualification for SLK, although transplant centers may choose to proceed with an LTA for other reasons. This study provides critical insight into the new SLK policy’s early effects on waitlist and post-transplant outcomes in patients listed for SLK and recipients despite these limitations that are related to the OPTN/UNOS registry.
Conclusions
This study showed that, following the implementation of the 2017 OPTN/UNOS policy, although the number of patient registrations increased, the number of SLK transplants decreased, and the number of liver transplants increased. Liver transplantation in this patient cohort was associated with worse post-transplant outcomes. These findings are consistent with previous studies and may have implications for future modifications to the current transplant policy
Figures
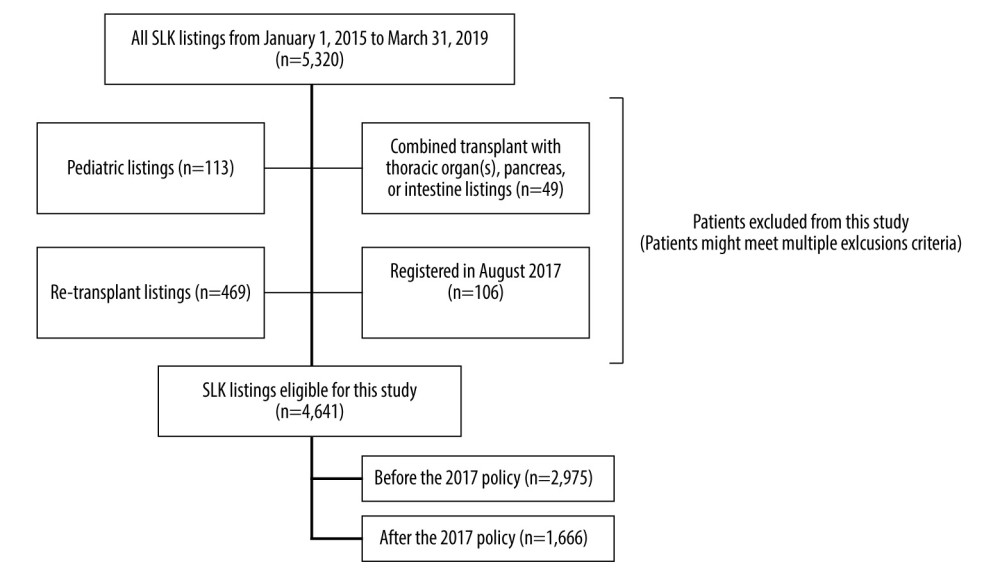 Figure 1. Figure explains the selection criteria of the study cohort. A total of 4641 of the 5320 patients who were listed for simultaneous liver-kidney transplant (SLK) between January 2015 and March 2019 were eligible for this study. Patients were divided into 2 groups: before the 2017 policy (n=2975), and after the 2017 policy (n=1666).
Figure 1. Figure explains the selection criteria of the study cohort. A total of 4641 of the 5320 patients who were listed for simultaneous liver-kidney transplant (SLK) between January 2015 and March 2019 were eligible for this study. Patients were divided into 2 groups: before the 2017 policy (n=2975), and after the 2017 policy (n=1666). 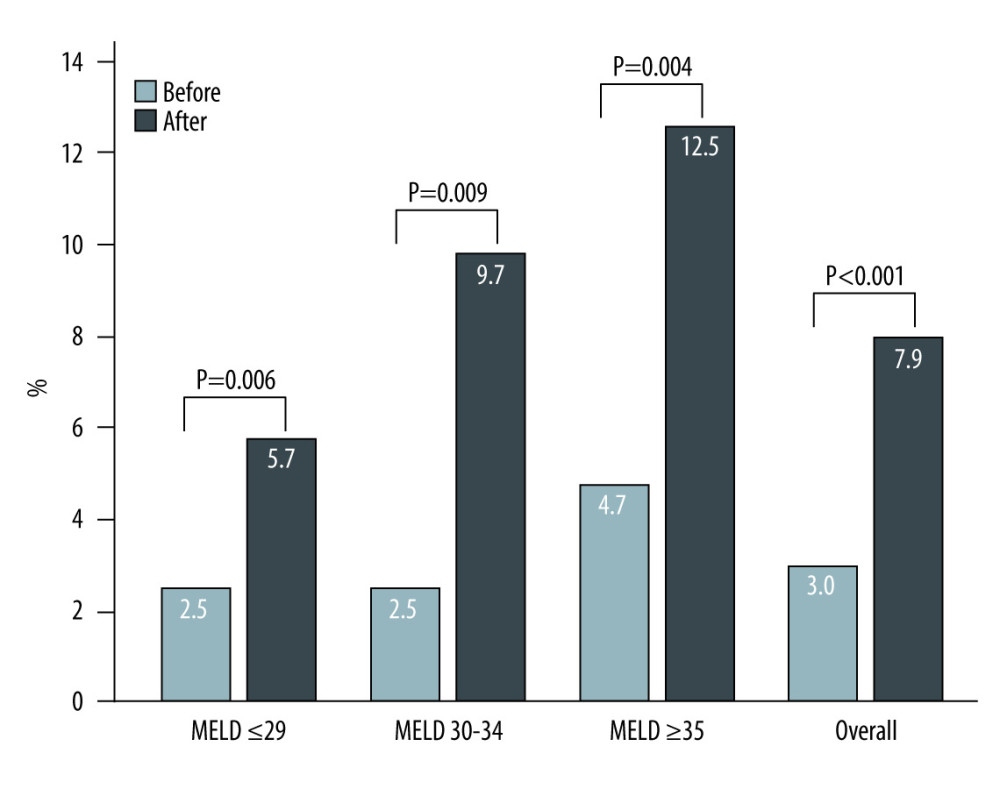 Figure 2. The proportion of liver transplantation alone (LTA) in simultaneous liver-kidney transplantation (SLK) listings increased in after the 2017 policy (3.0% in before the 2017 policy vs 7.9% in after the 2017 policy, P<0.001). Among each model for end-stage liver disease (MELD) score group, similar trends were observed (MELD score ≤29: 5.7% vs 2.5%, P=0.006; MELD score 30–34: 9.7% vs 2.5%, P=0.009; MELD score ≥35: 12.5% vs 4.7%, P=0.004). Light solid bar: LTA before the 2017 policy. Dark solid bar: LTA after the 2017 policy.
Figure 2. The proportion of liver transplantation alone (LTA) in simultaneous liver-kidney transplantation (SLK) listings increased in after the 2017 policy (3.0% in before the 2017 policy vs 7.9% in after the 2017 policy, P<0.001). Among each model for end-stage liver disease (MELD) score group, similar trends were observed (MELD score ≤29: 5.7% vs 2.5%, P=0.006; MELD score 30–34: 9.7% vs 2.5%, P=0.009; MELD score ≥35: 12.5% vs 4.7%, P=0.004). Light solid bar: LTA before the 2017 policy. Dark solid bar: LTA after the 2017 policy. 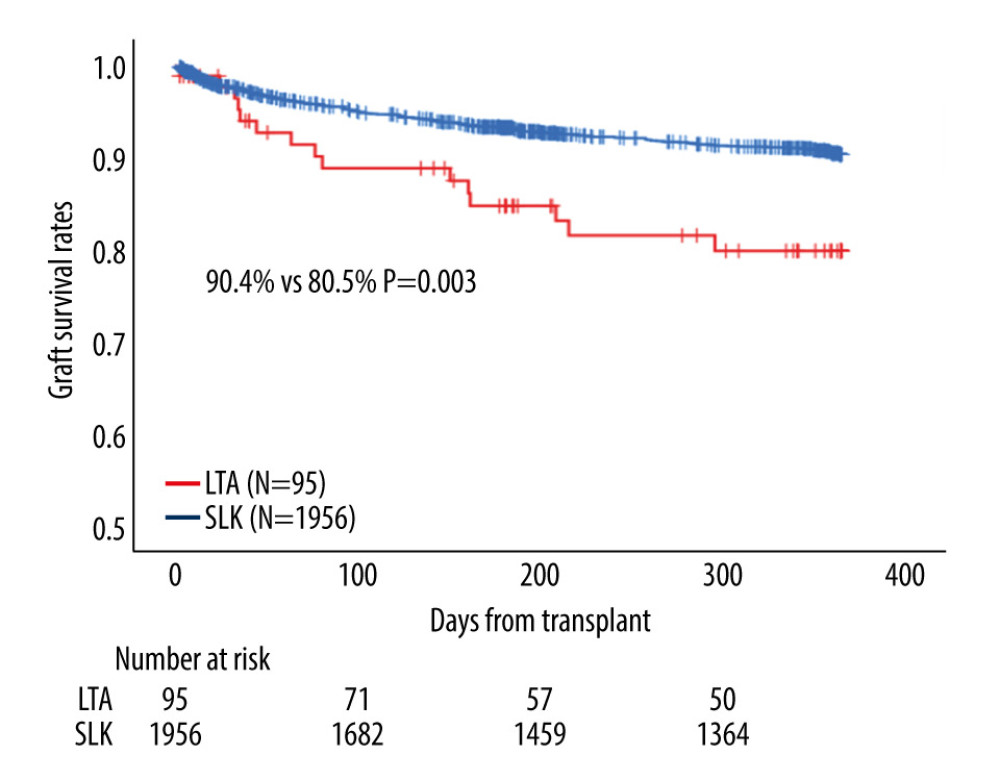 Figure 3. Among patients on simultaneous liver-kidney transplantation (SLK) listings, patients who received liver transplant alone (LTA) showed a significantly worse 1-year graft survival rate than those with SLK in all eras (80.5% vs 90.4%, P=0.003).
Figure 3. Among patients on simultaneous liver-kidney transplantation (SLK) listings, patients who received liver transplant alone (LTA) showed a significantly worse 1-year graft survival rate than those with SLK in all eras (80.5% vs 90.4%, P=0.003). 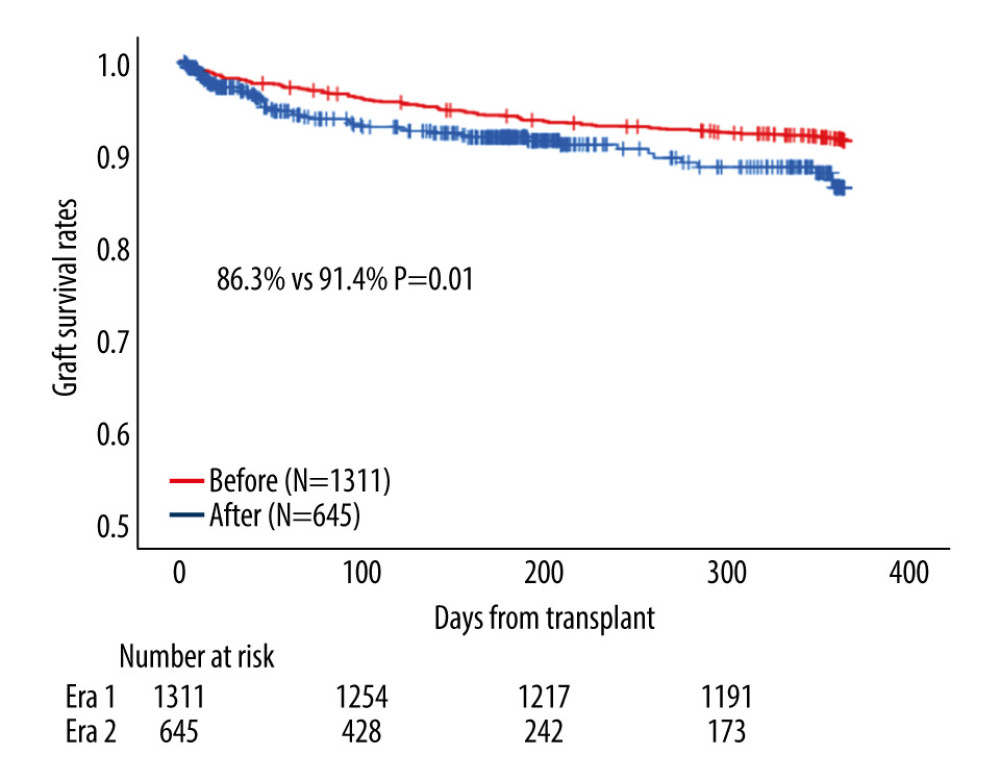 Figure 4. Among patients who received simultaneous liver-kidney transplantation (SLK), patients in after the 2017 policy showed a significantly worse 1-year graft survival rate than did patients before the 2017 policy (86.3% vs 91.4%, P=0.01).
Figure 4. Among patients who received simultaneous liver-kidney transplantation (SLK), patients in after the 2017 policy showed a significantly worse 1-year graft survival rate than did patients before the 2017 policy (86.3% vs 91.4%, P=0.01). 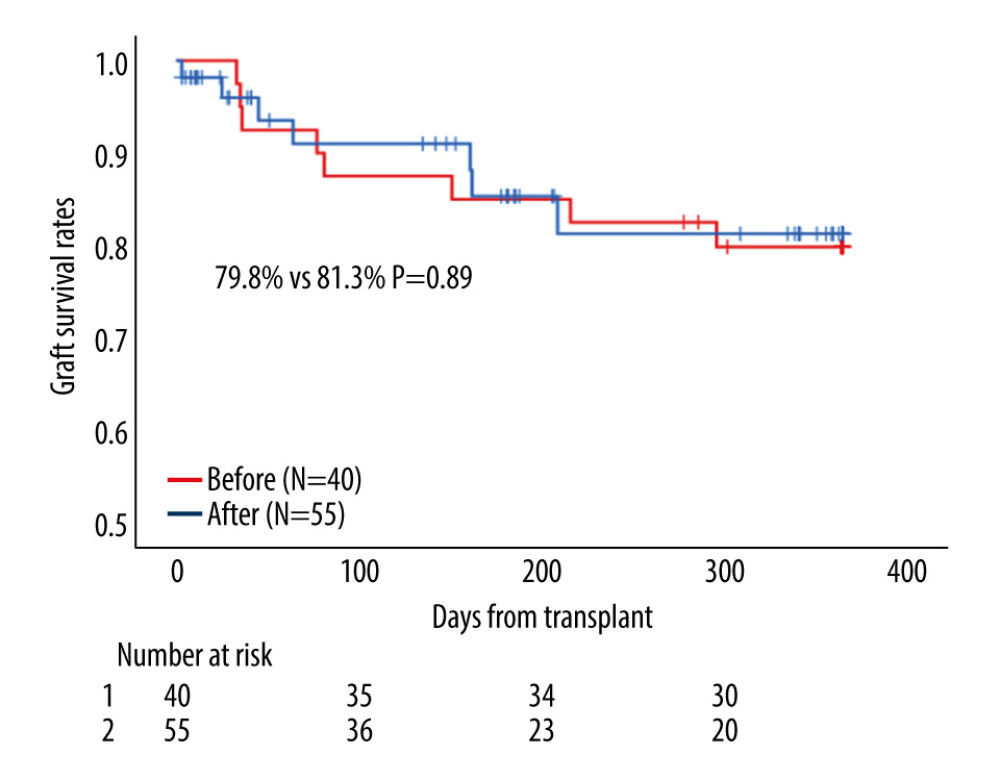 Figure 5. Among patients who received liver transplantation alone (LTA), patients after the 2017 policy showed a similar 1-year graft survival rate than did patients before the 2017 policy (81.3% vs 79.8%, P=0.89).
Figure 5. Among patients who received liver transplantation alone (LTA), patients after the 2017 policy showed a similar 1-year graft survival rate than did patients before the 2017 policy (81.3% vs 79.8%, P=0.89). Tables
Table 1. Patient characteristics in simultaneous liver-kidney transplantation listings before and after the 2017 policy.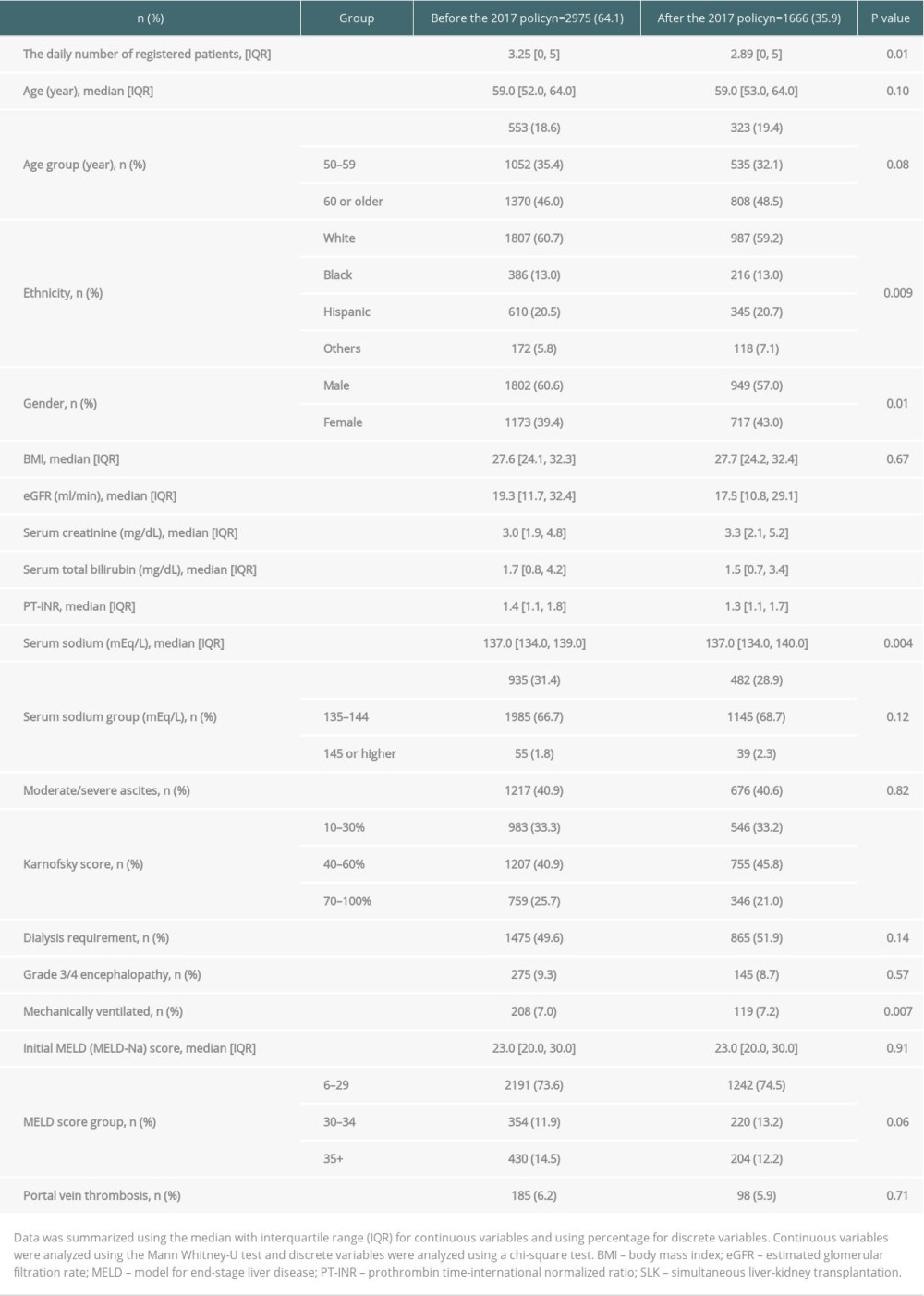 Table 2. The 90-day waitlist mortality risk and transplant probability after the 2017 policy (ref. before the 2017 policy).
Table 2. The 90-day waitlist mortality risk and transplant probability after the 2017 policy (ref. before the 2017 policy).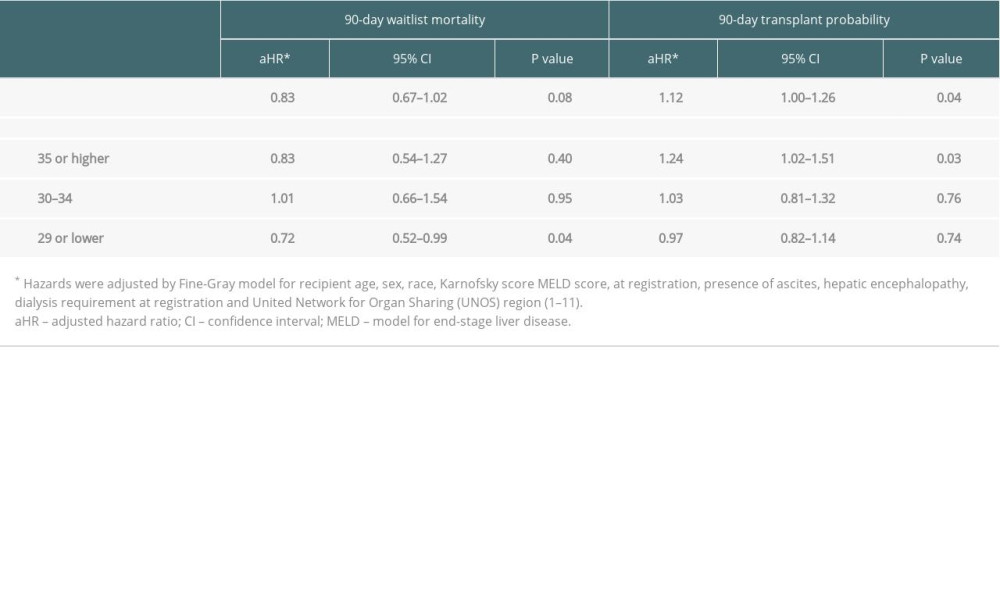 Table 3. Transplanted patient characteristics according to the final transplant type in patients who were initially listed for simultaneous liver-kidney transplantation.
Table 3. Transplanted patient characteristics according to the final transplant type in patients who were initially listed for simultaneous liver-kidney transplantation.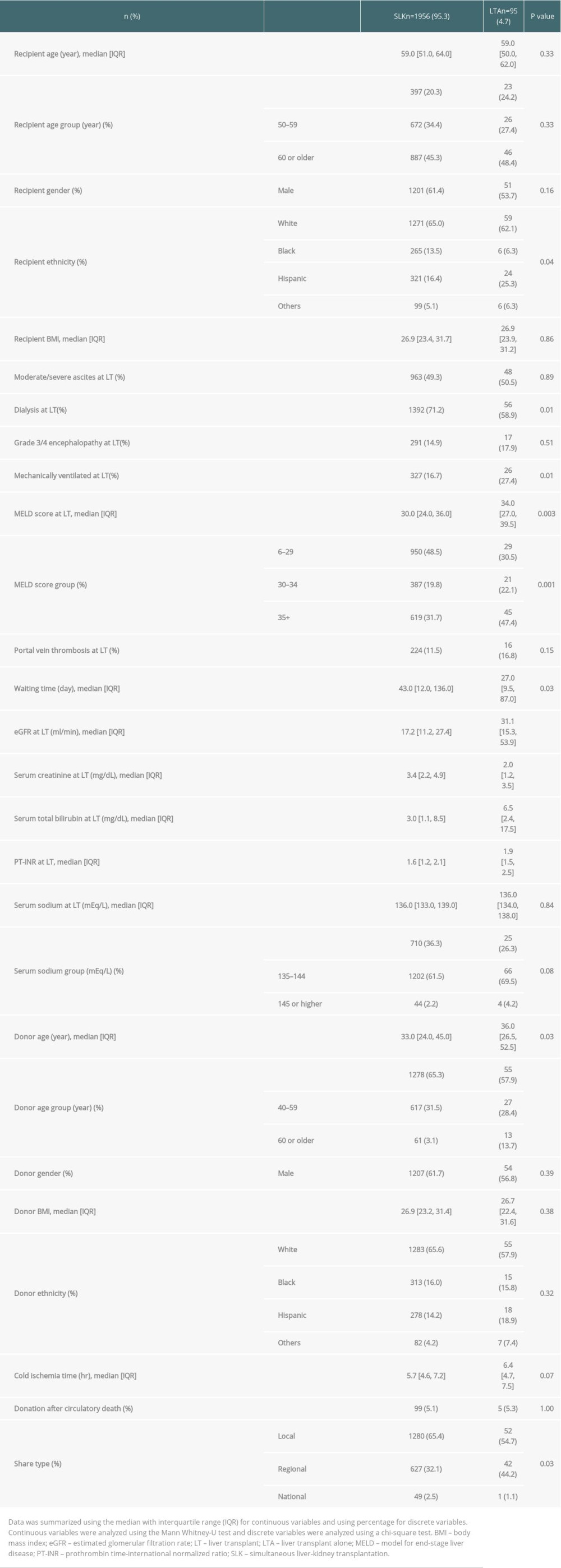 Table 4. Risk of 1-year graft loss and mortality after liver transplant alone in patients initially registered for simultaneous liver-kidney transplantation (ref. SLK recipients).
Table 4. Risk of 1-year graft loss and mortality after liver transplant alone in patients initially registered for simultaneous liver-kidney transplantation (ref. SLK recipients).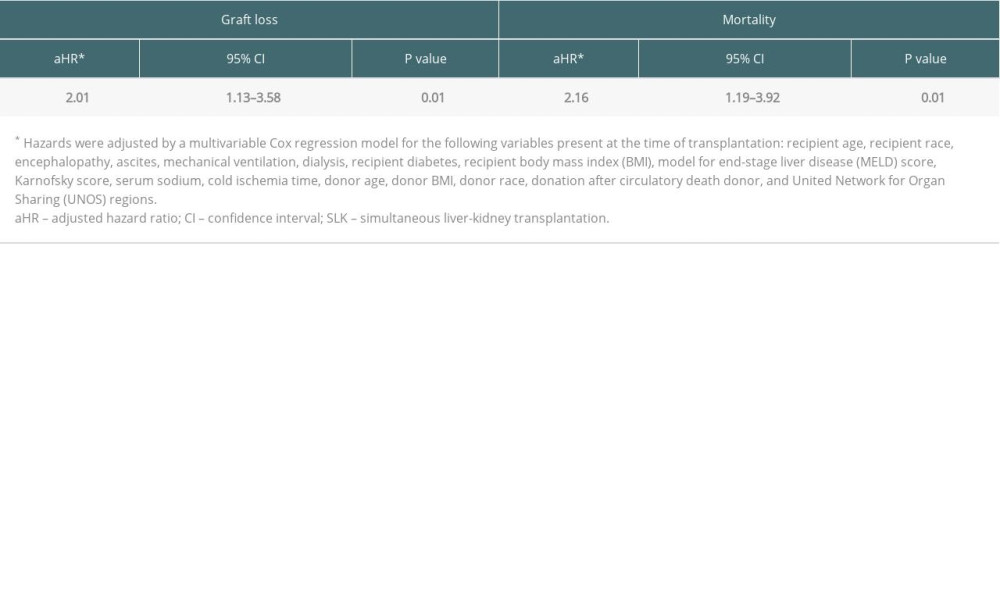 Table 5. Transplanted patient characteristics at time of simultaneous liver-kidney transplantation between the eras.
Table 5. Transplanted patient characteristics at time of simultaneous liver-kidney transplantation between the eras.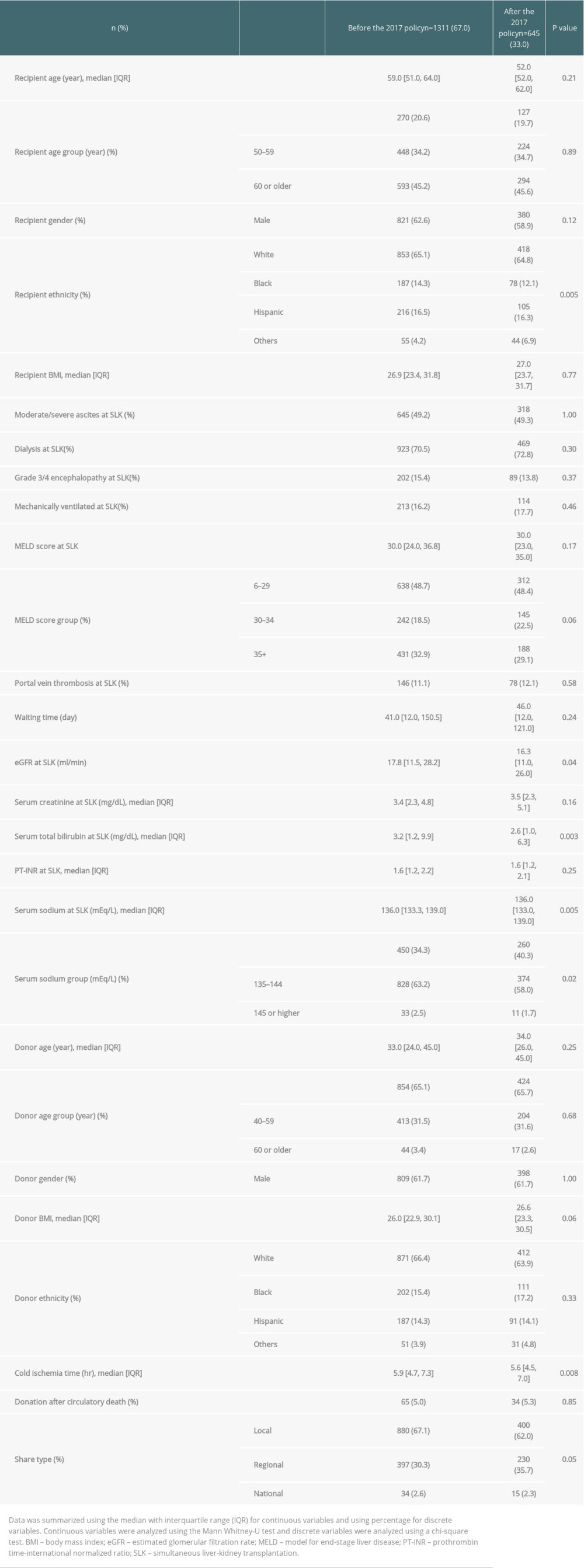 Table 6. Risk factors of 1-year graft loss and mortality after simultaneous liver-kidney transplantation.
Table 6. Risk factors of 1-year graft loss and mortality after simultaneous liver-kidney transplantation.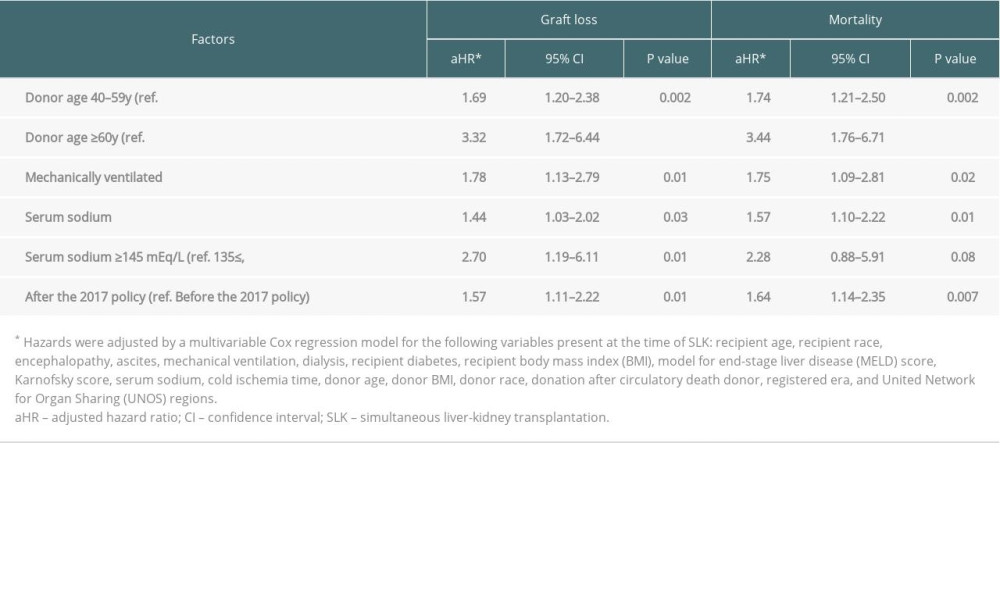 Table 7. Cause of 1-year death in patient who underwent simultaneous liver-kidney transplantation between the eras.
Table 7. Cause of 1-year death in patient who underwent simultaneous liver-kidney transplantation between the eras.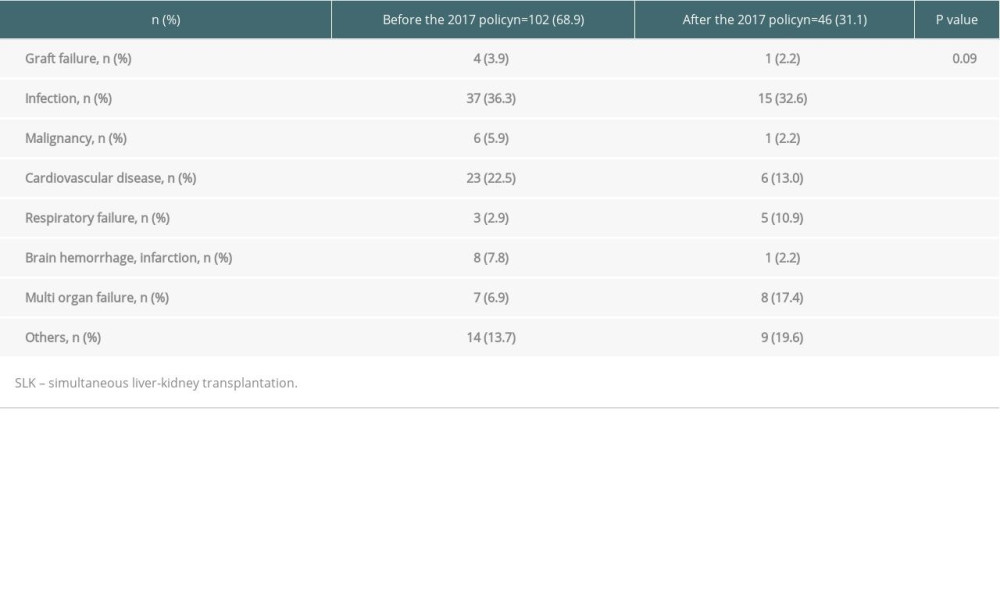 Table 8. Transplanted patient characteristics at time of liver transplant alone between the eras.
Table 8. Transplanted patient characteristics at time of liver transplant alone between the eras.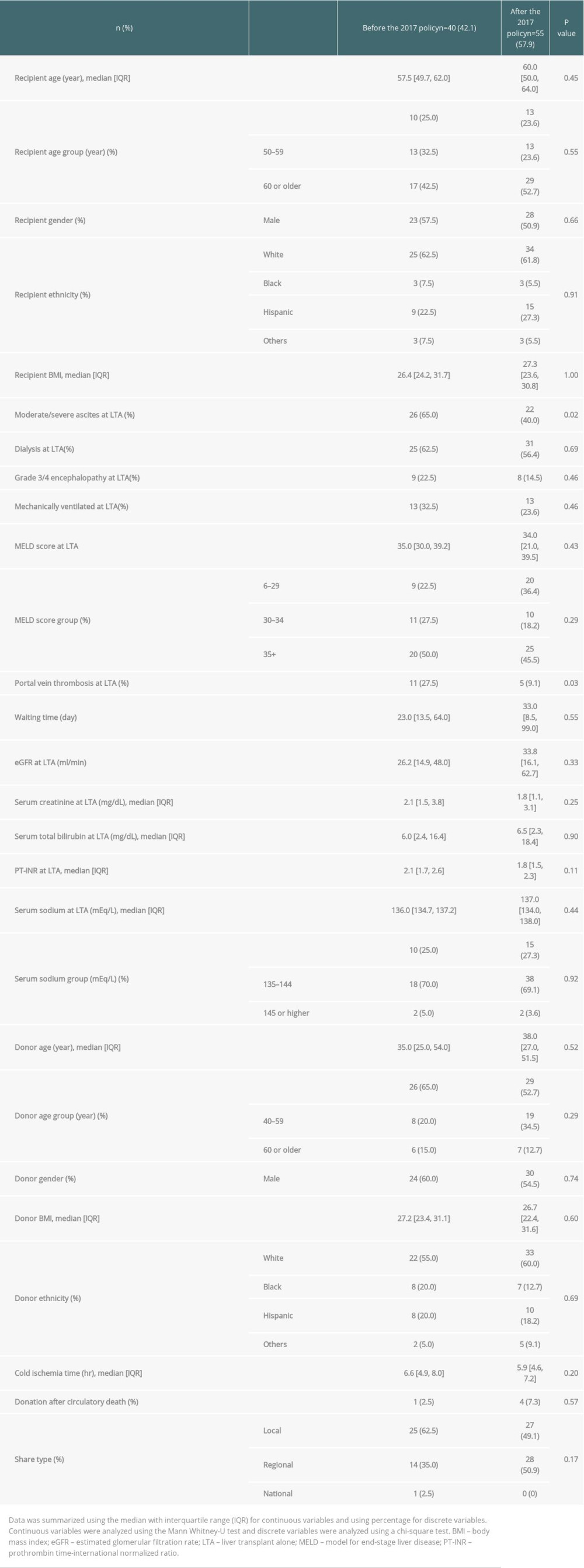 Table 9. Cause of 1-year death in patient who underwent liver transplant alone between the eras.
Table 9. Cause of 1-year death in patient who underwent liver transplant alone between the eras.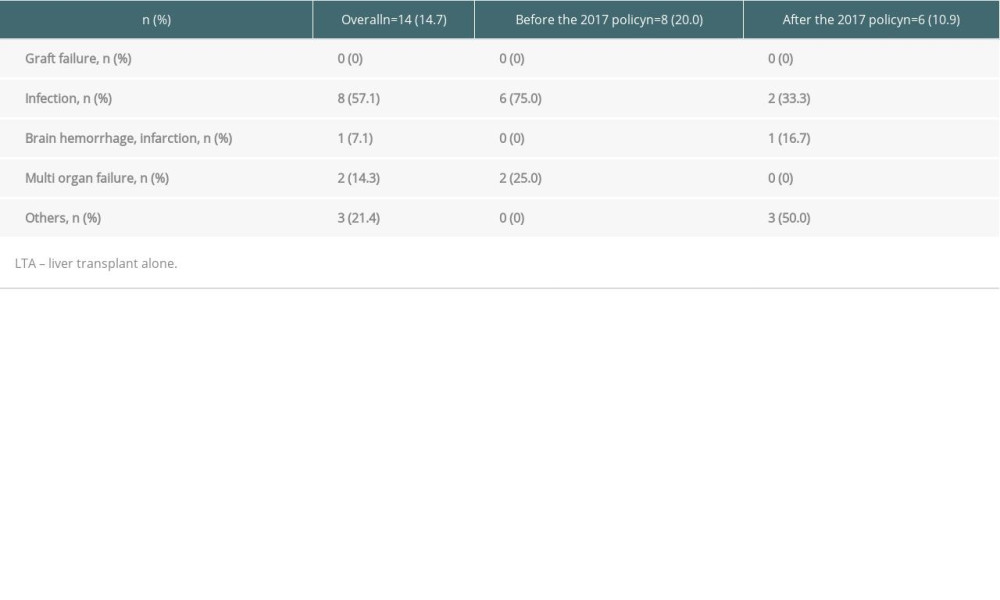
References
1. O’Leary JG, Levitsky J, Wong F, Protecting the kidney in liver transplant candidates: practice-based recommendations from the American Society of Transplantation Liver and Intestine Community of Practice: Am J Transplant, 2016; 16; 2516-31
2. Singal AK, Ong S, Satapathy SK, Simultaneous liver kidney transplantation: Transpl Int, 2019; 32; 343-52
3. Kim WR, Lake JR, Smith JM, OPTN/SRTR 2015 annual data report: Liver: Am J Transplant, 2017; 17(Suppl 1); 174-251
4. Miles CD, Westphal S, Liapakis A, Formica R, Simultaneous liver-kidney transplantation: Impact on liver transplant patients and the kidney transplant waiting list: Curr Transplant Rep, 2018; 5; 1-6
5. Formica RN, Aeder M, Boyle G, Simultaneous liver-kidney allocation policy: A proposal to optimize appropriate utilization of scarce resources: Am J Transplant, 2016; 16; 758-66
6. Nagai S, Safwan M, Collins K, Liver alone or simultaneous liver – kidney transplant? Pretransplant chronic kidney disease and post-transplant outcome – a retrospective study: Transpl Int, 2018; 31; 1028-40
7. : Organ Procurement and Transplant Network/United Network for Organ Sharing Kidney Transplantation Committee Simultaneous Liver Kidney (SLK) Allocation Policy, 2017 [Cited 2021 Dec 12]. Available from: https://optn.transplant.hrsa.gov/media/1192/0815-12_slk_allocation.pdf#:~:text=current%20policy%3A%20The%20current%20policy%20for%20SLK%20allocation,to%20ensure%20equity%20and%20efficiency%20in%20the%20U.S
8. Cullaro G, Hirose R, Lai JC, Changes in simultaneous liver-kidney transplant allocation policy may impact post liver transplant outcomes: Transplantation, 2019; 103; 959-64
9. Wilk AR, Booker SE, Stewart DE, Developing simultaneous liver-kidney transplant medical eligibility criteria while providing a safety net: A 2-year review of the OPTN’s allocation policy: Am J Transplant, 2021; 21; 3593-607
10. Nagai S, Suzuki Y, Kitajima T, Paradigm change in liver transplantation practice after the implementation of the liver-kidney allocation policy: Liver Transpl, 2021; 27; 1563-76
11. Gray RJ, A class of K-sample tests for comparing the cumulative incidence of a competing risk: Ann Stat, 1988; 16; 1141-54
12. Fine PF, Gray RJ, A proportional hazards model for the subdistribution of a competing risk: J Am Stat Assoc, 1999; 94; 496-509
13. Bahirwani R, Campbell MS, Siropaides T, Transplantation: Impact of pretransplant renal insufficiency: Liver Transpl, 2008; 14; 665-71
14. Jay CL, Washburn WK, Rogers J, Difference in survival in early kidney after liver transplantation compared with simultaneous liver-kidney transplantation: Evaluating the potential of the “Safety Net”: J Am Coll Surg, 2020; 230; 463-73
15. Samoylova ML, Wegermann K, Shaw BI, The impact of the 2017 kidney allocation policy change on simultaneous liver kidney utilization and outcomes: Liver Transpl, 2021; 27; 1106-15
16. Organ Procurement and Transplantation Network Liver and Intestine Committee: Allocation of livers and liver intestines [Cited 2021 Jun 8]. Available from: https://optn.transplant.hrsa.gov/media/eavh5bf3/optn_policies.pdf
17. Nagai S, Lucy CC, Schilke RE, Effects of allocating livers for transplantation based on model for end-stage liver disease-sodium scores on patient outcomes: Gastroenterology, 2018; 155; 1451-62
18. Ruf AE, Kremers WK, Chavez LL, Addition of serum sodium into the MELD score predicts waiting list mortality better than MELD alone: Liver Transpl, 2005; 11; 336-43
19. Biggins SW, Kim WR, Terrault NA, Evidence-based incorporation of serum sodium concentration into MELD: Gastroenterology, 2006; 130; 1652-60
20. Ismail MS, Hassan M, Martinez-Camacho A, Retrospective analysis of long-term outcome 10 years after liver transplantation for Wilson disease: Experience over three decades: Transpl Int, 2020; 33; 925-35
21. Organ Procurement and Transplantation Network: Donors Recovered in the US by Donor Type- Donors Recovered: January 1, 1988-April 30, 2021. Available from: https://optn.transplant.hrsa.gov/data/view-data-reports/national-data/#
22. Wiseman AC, Simultaneous liver-kidney transplant: long-term management (steroid withdrawal and safety net patients): Clin Liver Dis, 2019; 13; 176-89
23. Thongprayoon C, Kaewput W, Thamcharoen N, Incidence and impact of acute kidney injury after liver transplantation: A meta-analysis: J Clin Med, 2019; 8; 372
Figures
 Figure 1. Figure explains the selection criteria of the study cohort. A total of 4641 of the 5320 patients who were listed for simultaneous liver-kidney transplant (SLK) between January 2015 and March 2019 were eligible for this study. Patients were divided into 2 groups: before the 2017 policy (n=2975), and after the 2017 policy (n=1666).
Figure 1. Figure explains the selection criteria of the study cohort. A total of 4641 of the 5320 patients who were listed for simultaneous liver-kidney transplant (SLK) between January 2015 and March 2019 were eligible for this study. Patients were divided into 2 groups: before the 2017 policy (n=2975), and after the 2017 policy (n=1666). Figure 2. The proportion of liver transplantation alone (LTA) in simultaneous liver-kidney transplantation (SLK) listings increased in after the 2017 policy (3.0% in before the 2017 policy vs 7.9% in after the 2017 policy, P<0.001). Among each model for end-stage liver disease (MELD) score group, similar trends were observed (MELD score ≤29: 5.7% vs 2.5%, P=0.006; MELD score 30–34: 9.7% vs 2.5%, P=0.009; MELD score ≥35: 12.5% vs 4.7%, P=0.004). Light solid bar: LTA before the 2017 policy. Dark solid bar: LTA after the 2017 policy.
Figure 2. The proportion of liver transplantation alone (LTA) in simultaneous liver-kidney transplantation (SLK) listings increased in after the 2017 policy (3.0% in before the 2017 policy vs 7.9% in after the 2017 policy, P<0.001). Among each model for end-stage liver disease (MELD) score group, similar trends were observed (MELD score ≤29: 5.7% vs 2.5%, P=0.006; MELD score 30–34: 9.7% vs 2.5%, P=0.009; MELD score ≥35: 12.5% vs 4.7%, P=0.004). Light solid bar: LTA before the 2017 policy. Dark solid bar: LTA after the 2017 policy. Figure 3. Among patients on simultaneous liver-kidney transplantation (SLK) listings, patients who received liver transplant alone (LTA) showed a significantly worse 1-year graft survival rate than those with SLK in all eras (80.5% vs 90.4%, P=0.003).
Figure 3. Among patients on simultaneous liver-kidney transplantation (SLK) listings, patients who received liver transplant alone (LTA) showed a significantly worse 1-year graft survival rate than those with SLK in all eras (80.5% vs 90.4%, P=0.003). Figure 4. Among patients who received simultaneous liver-kidney transplantation (SLK), patients in after the 2017 policy showed a significantly worse 1-year graft survival rate than did patients before the 2017 policy (86.3% vs 91.4%, P=0.01).
Figure 4. Among patients who received simultaneous liver-kidney transplantation (SLK), patients in after the 2017 policy showed a significantly worse 1-year graft survival rate than did patients before the 2017 policy (86.3% vs 91.4%, P=0.01). Figure 5. Among patients who received liver transplantation alone (LTA), patients after the 2017 policy showed a similar 1-year graft survival rate than did patients before the 2017 policy (81.3% vs 79.8%, P=0.89).
Figure 5. Among patients who received liver transplantation alone (LTA), patients after the 2017 policy showed a similar 1-year graft survival rate than did patients before the 2017 policy (81.3% vs 79.8%, P=0.89). Tables
 Table 1. Patient characteristics in simultaneous liver-kidney transplantation listings before and after the 2017 policy.
Table 1. Patient characteristics in simultaneous liver-kidney transplantation listings before and after the 2017 policy. Table 2. The 90-day waitlist mortality risk and transplant probability after the 2017 policy (ref. before the 2017 policy).
Table 2. The 90-day waitlist mortality risk and transplant probability after the 2017 policy (ref. before the 2017 policy). Table 3. Transplanted patient characteristics according to the final transplant type in patients who were initially listed for simultaneous liver-kidney transplantation.
Table 3. Transplanted patient characteristics according to the final transplant type in patients who were initially listed for simultaneous liver-kidney transplantation. Table 4. Risk of 1-year graft loss and mortality after liver transplant alone in patients initially registered for simultaneous liver-kidney transplantation (ref. SLK recipients).
Table 4. Risk of 1-year graft loss and mortality after liver transplant alone in patients initially registered for simultaneous liver-kidney transplantation (ref. SLK recipients). Table 5. Transplanted patient characteristics at time of simultaneous liver-kidney transplantation between the eras.
Table 5. Transplanted patient characteristics at time of simultaneous liver-kidney transplantation between the eras. Table 6. Risk factors of 1-year graft loss and mortality after simultaneous liver-kidney transplantation.
Table 6. Risk factors of 1-year graft loss and mortality after simultaneous liver-kidney transplantation. Table 7. Cause of 1-year death in patient who underwent simultaneous liver-kidney transplantation between the eras.
Table 7. Cause of 1-year death in patient who underwent simultaneous liver-kidney transplantation between the eras. Table 8. Transplanted patient characteristics at time of liver transplant alone between the eras.
Table 8. Transplanted patient characteristics at time of liver transplant alone between the eras. Table 9. Cause of 1-year death in patient who underwent liver transplant alone between the eras.
Table 9. Cause of 1-year death in patient who underwent liver transplant alone between the eras. Table 1. Patient characteristics in simultaneous liver-kidney transplantation listings before and after the 2017 policy.
Table 1. Patient characteristics in simultaneous liver-kidney transplantation listings before and after the 2017 policy. Table 2. The 90-day waitlist mortality risk and transplant probability after the 2017 policy (ref. before the 2017 policy).
Table 2. The 90-day waitlist mortality risk and transplant probability after the 2017 policy (ref. before the 2017 policy). Table 3. Transplanted patient characteristics according to the final transplant type in patients who were initially listed for simultaneous liver-kidney transplantation.
Table 3. Transplanted patient characteristics according to the final transplant type in patients who were initially listed for simultaneous liver-kidney transplantation. Table 4. Risk of 1-year graft loss and mortality after liver transplant alone in patients initially registered for simultaneous liver-kidney transplantation (ref. SLK recipients).
Table 4. Risk of 1-year graft loss and mortality after liver transplant alone in patients initially registered for simultaneous liver-kidney transplantation (ref. SLK recipients). Table 5. Transplanted patient characteristics at time of simultaneous liver-kidney transplantation between the eras.
Table 5. Transplanted patient characteristics at time of simultaneous liver-kidney transplantation between the eras. Table 6. Risk factors of 1-year graft loss and mortality after simultaneous liver-kidney transplantation.
Table 6. Risk factors of 1-year graft loss and mortality after simultaneous liver-kidney transplantation. Table 7. Cause of 1-year death in patient who underwent simultaneous liver-kidney transplantation between the eras.
Table 7. Cause of 1-year death in patient who underwent simultaneous liver-kidney transplantation between the eras. Table 8. Transplanted patient characteristics at time of liver transplant alone between the eras.
Table 8. Transplanted patient characteristics at time of liver transplant alone between the eras. Table 9. Cause of 1-year death in patient who underwent liver transplant alone between the eras.
Table 9. Cause of 1-year death in patient who underwent liver transplant alone between the eras. In Press
18 Mar 2024 : Original article
Does Antibiotic Use Increase the Risk of Post-Transplantation Diabetes Mellitus? A Retrospective Study of R...Ann Transplant In Press; DOI: 10.12659/AOT.943282
20 Mar 2024 : Original article
Transplant Nephrectomy: A Comparative Study of Timing and Techniques in a Single InstitutionAnn Transplant In Press; DOI: 10.12659/AOT.942252
28 Mar 2024 : Original article
Association Between FEV₁ Decline Rate and Mortality in Long-Term Follow-Up of a 21-Patient Pilot Clinical T...Ann Transplant In Press; DOI: 10.12659/AOT.942823
02 Apr 2024 : Original article
Liver Transplantation from Brain-Dead Donors with Hepatitis B or C in South Korea: A 2014-2020 Korean Organ...Ann Transplant In Press; DOI: 10.12659/AOT.943588
Most Viewed Current Articles
05 Apr 2022 : Original article
Impact of Statins on Hepatocellular Carcinoma Recurrence After Living-Donor Liver TransplantationDOI :10.12659/AOT.935604
Ann Transplant 2022; 27:e935604
12 Jan 2022 : Original article
Risk Factors for Developing BK Virus-Associated Nephropathy: A Single-Center Retrospective Cohort Study of ...DOI :10.12659/AOT.934738
Ann Transplant 2022; 27:e934738
22 Nov 2022 : Original article
Long-Term Effects of Everolimus-Facilitated Tacrolimus Reduction in Living-Donor Liver Transplant Recipient...DOI :10.12659/AOT.937988
Ann Transplant 2022; 27:e937988
15 Mar 2022 : Case report
Combined Liver, Pancreas-Duodenum, and Kidney Transplantation for Patients with Hepatitis B Cirrhosis, Urem...DOI :10.12659/AOT.935860
Ann Transplant 2022; 27:e935860








