23 April 2021: Original Paper
A Single-Center Experience with Lung Transplants During the COVID-19 Pandemic
Wojtek Karolak1ABE, Elżbieta Woźniak-Grygiel2CE, Monika Łącka3BC, Jacek Wojarski1BG, Salma Ali Addo4EF, Adith Kumaravel4EF, Salin Khan4EF, Reema Shinde4EF, Ikram Mukhtar Nuur4EF, Feven Kifle Reta4EF, Jan Rogowski1G, Ewa Jassem4EG, Krystyna Pastwa4F, Aleksandra Pałasz1B, Nikodem Ulatowski1B, Sławomir Żegleń24ADG*DOI: 10.12659/AOT.929946
Ann Transplant 2021; 26:e929946
Abstract
BACKGROUND: This single-center study analyzed distinctions between lung transplants performed in the Department of Cardiac and Vascular surgery of the University Clinical Center in Gdańsk, Poland before and during the COVID-19 pandemic.
MATERIAL AND METHODS: There were 189 patients who underwent the qualification procedure to lung transplantation in the Department of Cardiac and Vascular Surgery of the University Clinical Center in Gdańsk, Poland in the years 2019 and 2020. The control group consisted of 12 patients transplanted in 2019, and the study group consisted of 16 patients transplanted in 2020.
RESULTS: During 2019, the qualification process was performed in 102 patients with pulmonary end-stage diseases. In 2020, despite the 3-month lockdown related to organizational changes in the hospital, 87 qualification processes were performed. The mortality rate of patients on the waiting list in 2020 was 14.3% (6 patients died), and during 2019 the rate was also 14.3% (4 patients died). Donor qualifications were according to ISHLT criteria. The distribution of donors in both years was similar. There was no relationship between the geographic area of residence and source of donors. In 2019, all 12 patients had double-lung transplant. In 2020, 11 patients had double-lung transplant and 5 patients had single-lung transplant. There was no difference in ventilation time and PGD aside from a shorter ICU stay in 2020.
CONCLUSIONS: Lung transplants were relatively well-conducted despite the continued obstacles of the COVID-19 pandemic.
Keywords: COVID-19, Lung Transplantation, Pandemics, COVID-19, Follow-Up Studies, Health Services Accessibility, Outcome and Process Assessment, Health Care, Poland, Tissue and Organ Procurement, Waiting Lists
Background
The rapidly growing number of COVID-19 deaths, as well as infected patients, along with the significant stress that it adds on healthcare systems, is a devastating situation we are living in during this pandemic. As of today 31 October 2020, there have been more than 46 million cases worldwide [1] and more than 360 000 cases confirmed in Poland [2].
The SARS-CoV-2 coronavirus was first discovered in the city of Wuhan, Hubei Province, Central China, and in the past few months it has spread to 218 countries [3]. In December 2019, the first case was reported and an array of patients were admitted to hospital with initial diagnosis of pneumonia with unknown etiology.
Lung transplants have been established as an effective treatment for patients with end-stage respiratory failure unresponsive to other medical or surgical interventions [4] and still remains the treatment of choice for these patients. Annually, nearly 5000 lung transplants are performed around the world.
During the novel coronavirus pandemic, studies revealed a worldwide reduction in the number of lung transplants performed. Despite these substantially more difficult conditions, life-saving lung transplantations are still performed across the globe.
In all leading solid-organ transplant centers during the pandemic, the RT-PCR test is used preoperatively, preferably from a nasopharyngeal swab, although broncho-alveolar lavage was initially thought to be better [7]. In an international survey done by Benjamin Coiffard et al, 81% of the participating centers saw a reduction in lung transplant activity [8]. In the beginning, data from the Netherlands showed a significant decrease in the number of lung transplants [9], whereas Germany, with the highest AI (Accessibility Index), reported only a moderate decrease in lung transplant activity [10].
Notably, in the Eurotransplant countries, 1153 deceased-donor lungs were used for transplant during 2019 and in 2020, and 1077 lungs were used between January to October. Moreover, 1375 lung transplants in total were done in 2019. To continue their work, each member country follows preventive screening protocols [11].
Across the Atlantic, mid-pandemic reports from the U.S. clearly show a decrease in all solid-organ transplants, with kidney transplants decreasing the most, followed by livers, lungs, and hearts [12].
The aim of the present study was to analyze the impact of the pandemic on the course of the lung transplant program at the University Clinical Center in Gdańsk, Poland.
Material and Methods
STATISTICAL ANALYSIS:
Statistical analysis was performed with Microsoft Office Professional Plus 2013 (Microsoft Corporation) and Statistica 13.3 (StatSoft, Inc.). To assess the distribution, the Shapiro-Wilk test for normality was applied. Data are presented as mean±standard deviation or median±quartile deviation. The differences between means with
Results
QUALIFICATIONS FOR LUNG TRANSPLANTATION PROCEDURES IN 2019 AND 2020 (UNTIL NOVEMBER):
During 2019, the qualification process was performed in 102 patients with pulmonary end-stage diseases. In 2020, despite the 3-month lockdown related to organizational changes in the hospital, 87 qualifications processes were performed. Extrapolating, it seems that the number of qualifications conducted in 2019 and 2020 were be comparable. Detailed data on qualifications in individual years are presented in Table 1. In our center, the recipient list at the end of 2019 had 28 recipients and by the end of September 2020 it had 42. The mortality rate on the waiting list during 2020 was 14.3% (6 patients died), and during 2019 it was also 14.3% (4 patients died).
To date, 4 of the qualified patients have been infected with SARS-CoV-2, 2 of which were on the active waiting list and still remain on it (convalescents).
DEMOGRAPHIC INFORMATION OF TRANSPLANTED PATIENTS:
A group of 28 patients was selected, consisting of 10 women and 18 men. There were 6 women and 10 men in the study group (transplants in 2020), and 4 women and 8 men in the control group (transplants in 2019). The average age of the patients in the study group was 50 years, and 53.6 years in the control group (Table 2).
TYPE OF DISEASE:
The diseases that caused respiratory failure were chronic obstructive pulmonary disease (COPD) in 8 cases in the control group and 5 in the study group, cystic fibrosis (CF) in 1 case in the control group and 2 in the study group, idiopathic pulmonary arterial hypertension (IPAH) in 1 patient in the control group 2019 and 3 patients in the study group, extrinsic allergic alveolitis (EAA) in 1 patient in the study group, usual interstitial pneumonia (UIP) in 2 patients in study group, and idiopathic pulmonary fibrosis (IPF) in 2 cases from both groups. Three patients from both groups had concomitant pulmonary hypertension (HA). In 1 case in the study group, the cause of respiratory failure was COVID-19 related fibrosis.
SURGERY:
Donor qualifications were according to the ISHLT criteria. The geographic areas of residence of donors were comparable in both years. The distribution of donors in both years were similar; northern Poland dominated in 2019, with 8 donors from the north and 4 from the south, and in 2020 there were 11 patients from the north and 5 from the south. There was no relationship between geographic area of residence and source of donors. In 2019, all 12 patients had double-lung transplants. In 2020, 11 patients had double-lung transplant and 5 patients had single-lung transplant. We performed 6 off-pump surgeries, 5 surgeries on ECMO, and 1 surgery which required conversion from off-pump to ECMO in 2019. In 2020, we performed 2 off-pump surgery, 2 surgeries with conversion from off-pump to ECMO, and 12 surgeries on ECMO. In our program, any patient other than IPAH is considered for transplant without ECMO. Then, cardiopulmonary intraoperative evaluation is performed with the main criterion being patient safety.
The transplant mode was on schedule except for the 2020 COVID transplant case, which was urgent. The urgency criteria in Poland are very strict. The LAS does not apply. The main urgent recipient criteria according to POLTRANSPLAT are as follows: ECMO in a patient up to 55 years old with standard criteria for the application of ECMO and patients on a ventilator in a hospital without the possibility of going home (ventilator therapy over 24 h, patient up to age 55 years, primary graft failure [PGD ≥3] up to 7 days after transplantation); the patient is then urgent to re-LTx.
There was no difference between 2019 vs 2020 in intubation time (21.2±5.2 vs 19.5±5.7, p>0.05), PGD 0 h (1.0±0.8 vs 2.0±1.0, p>0.05) and PGD 72 h (1.0±0.5 vs 2.0±0.76, p>0.05). ICU (intensive care unit) time was significantly shorter in 2020 in compared to 2019 (64.4±35.2 vs 155.4±94.2, p=0.0226; respectively).
SHORT- AND LONG-TERM FOLLOW-UP:
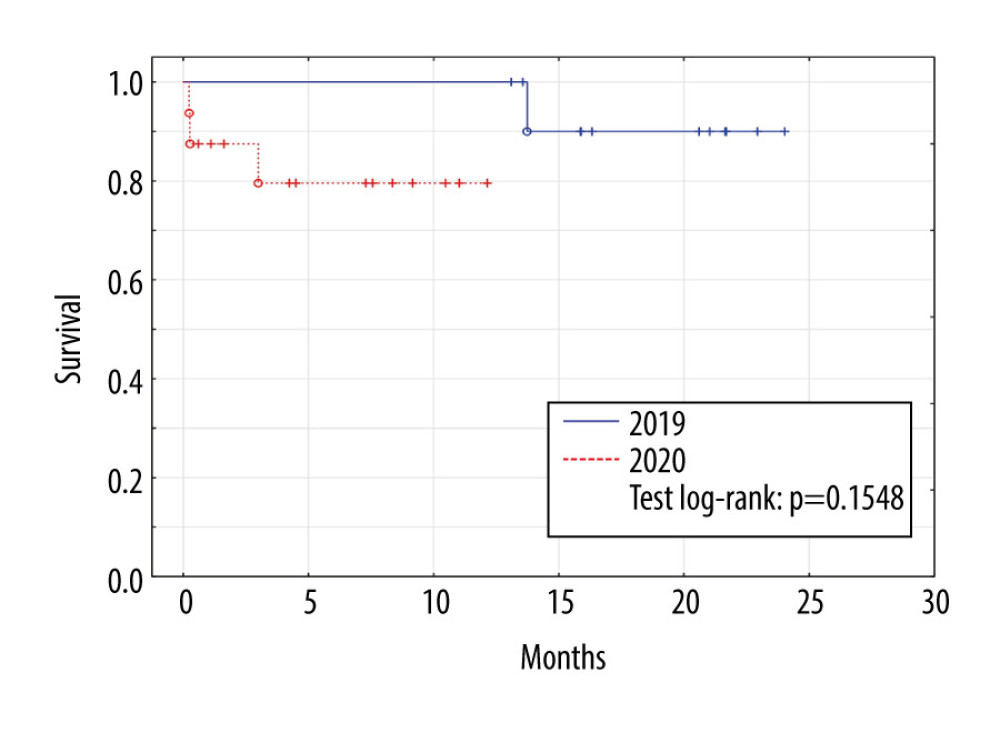
No patients in the control group (2019) died in 2019, but 1 from this group died in 2020 due to COVID-19. In the study group (2020), 3 patients died, 1 due to septic complications 3 months after transplant, 1 due to brain edema of undetermined cause in the immediate postoperative period, and 1 due to intracranial hemorrhage in the immediate postoperative period. No deaths of patients transplanted in 2020 were due to COVID-19-related causes, 1 patient transplanted in 2020 became ill due to COVID-19 infection, but the infection was mild and resulted in a full recovery (Figure 1).
Discussion
It should be emphasized that the lung transplant program in Gdańsk is under development. In its inception in 2018, 4 transplants were performed, and 12 were done in 2019.
It is made up of an experienced team, which had previously formed the largest transplant team in southern Poland. The development strategy had a steady annual increase in the number of transplants. It seems that COVID-19 had an impact on the qualification for transplantation, reducing the dynamics of program development in the form of a reduction in the assumed number of performed qualification processes. Data obtained by the Polish Transplant Coordinating Center (Poltransplant) webpage
Each month corresponds to patients waiting on the last day of a given month. The reports indicate a steady increase of patients in the waiting list as well as a small increase in the number of performed transplants. The average number of patients on the waiting list increased in 2020 compared to 2018 and 2019. Moreover, 9 more transplants were performed in 2019 than in 2020. It should be emphasized that in 2018 the pulmonary program in Zabrze was divided and a new center was opened in Gdańsk by part of an experienced team. The priority was to quickly create a list of recipients in the new center; hence, its nationwide growth. There was a 13.5% increase in patients on the waiting list between the beginning and end of 2020 compared to a 24% increase in 2019.
An important message of the study is that despite the tragic consequences of the pandemic, transplants are being routinely performed and that the introduced standards for organ procurement and transplantations are effective. One patient transplanted in 2019 died in 2020 due to COVID-19 (infected at home). Only 4 of the qualified patients were infected with COVID-19, and none of them were infected in the hospital (at the moment we have information about their recovery/survival). The most important procedures for recipient protection were obligatory donor swabbing, chest computed tomography for each donor, and buffer wards.
The International Society for Heart and Lung Transplantation (ISHLT) and American Society of Transplant Surgeons (ASTS) have issued guidelines for transplantation regarding immunosuppression and donor-derived infection in newly transplanted patients during the COVID-19 pandemic [13,14].
There is a general consensus between ASTS and ISHLT in the continuation of life-saving transplants while minimizing the risk of COVID-19 in patients waiting for transplant, hospitalized patients, and recipients. As there is no data on community-acquired COVID-19 and blood-acquired COVID-19, organ donation and recipients have to be carefully evaluated [13,14]. In addition, there is a general understanding of the contraindication of COVID-19-positive organ utilization. Precautions have to be taken for all people involved because of the limited data on the risk of rejection in immunosuppressed patients with COVID-19 infection. The immunocompromised patients are at a substantial risk and thus should avoid exposure by social distancing. Patients waiting for transplant should avoid visiting hospital settings and perform social distancing. Transplant recipients should acquire medication for longer periods of time as well as communicate with their transplant center in case of infection. In hospital settings, buffer wards are needed and limitations in visitors to prevent exposure. The transplant team needs to follow pertinent protocols such as carrying protective gear, performing proper hand hygiene, and social distancing.
ISHLT and ASTS provide continuous updates with the latest information and data providing answers to important questions about the pandemic.
Despite the availability of donor organs, fewer lung transplants have been performed. This is a consequence not only of the recent restrictions due to the pandemic, but also due to the uncertainty of suitability in severely ill patients with the risk of a COVID-19 infection. A decrease in transplants could also be attributed to a decrease in donor awareness among ICU and emergency physicians due to the added stressors of the pandemic [9].
Hospitals are forced to transform operating rooms into intensive care units. Utilizing these spaces for lung transplantation may be considered unethical due to limited time and resources.
It should be mentioned that the program has been adapted to the new pandemic conditions. The main points for donors are computed tomography (CT) examination of each, swabs (PCR), obligatory by POLTRANSPLANT, and analysis of the local epidemiological situation. The main points for recipients are swabs (PCR), buffer subunit until swab results, and complete isolation from COVID-19-positive patients (dedicated hospitals only for COVID-19-positive patients).
The procedure of lung transplantation per se faces challenges including recovery and assessment of deceased organ donors, postsurgical complications, transitional care to residence, and so forth. The COVID-19 pandemic imposes additional obstacles on the selection criteria of appropriate groups of patients. Nosocomial COVID-19 may also be a factor considered by patients or healthcare workers as an adequate reason for refraining from evaluating lung transplant eligibility [15].
Only a few groups have been reported to be performing lung transplantation successfully [16] in cases of COVID-19-associated ARDS. Nevertheless, these cases emphasize the importance of lung transplantation as yet another tool in the fight against this novel virus.
With this in mind, it is of great importance that patients who meet the criteria according to the traditional guidelines as well as the criteria set by the transplant center be considered for lung transplantation, regardless of the cause of irreversible lung damage.
Conclusions
Despite continuous new updates and information regarding this novel virus, much remains unknown. Further understanding is key in terms of risk versus benefit, patient ability to recover, the mechanism of lung injury in infected asymptomatic donors, and recipients’ risk of infection. Moreover, for the safety and long-term survival of these patients, more studies are needed on the timing, candidacy criteria, and management. The full impact on transplantation activities should be determined annually as the pandemic is still unfolding.
The results of this study should be interpreted with caution. This relatively low-volume lung transplant program works effectively in the conditions of the surrounding pandemic; this is the most important finding from our study. Of course, it is difficult to predict the future in today’s difficult times, but such information at the moment is of great importance for doctors and for patients who qualify for lung transplantation.
References
1. Johns Hopkins University & Medicine Coronavirus Resource Center: Home https://coronavirus.jhu.edu/
2. Johns Hopkins University & Medicine Coronavirus Resource Center: Poland – COVID-19 Overview https://coronavirus.jhu.edu/region/poland
3. : Coronavirus Update (Live): 46,726,237 Cases and 1,204,029 Deaths from COVID-19 Virus Pandemic – Worldometer https://www.worldometers.info/coronavirus/
4. Kotloff RM, Thabut G, Lung transplantation: Am J Respir Crit Care Med, 2011; 184(2); 159-71
5. Cypel M, Keshavjee S, When to consider lung transplantation for COVID-19: Lancet Respir Med, 2020; 8(10); 944-46
6. Lang C, Jaksch P, Hoda MA, Lung transplantation for COVID-19-associated acute respiratory distress syndrome in a PCR-positive patient: Lancet Respir Med, 2020; 8(10); 1057-60
7. Geri P, Salton F, Zuccatosta L, Limited role for bronchoalveolar lavage to exclude COVID-19 after negative upper respiratory tract swabs: A multicentre study: Eur Respir J, 2020; 56(4); 2001733
8. Coiffard B, Lepper PM, Prud’Homme E, Management of lung transplantation in the COVID-19 era – An international survey: Am J Transplant, 2020 [Online ahead of print]
9. de Vries APJ, Alwayn IPJ, Hoek RAS, Immediate impact of COVID-19 on transplant activity in the Netherlands: Transpl Immunol, 2020; 61; 101304
10. Michel S, Witt C, Gottlieb J, Aigner C, Impact of COVID-19 on lung transplant activity in Germany-A cross-sectional survey: Thorac Cardiovasc Surg, 2021; 69(1); 92-94
11. : Monthly statistics – Eurotransplant https://www.eurotransplant.org/statistics/monthly-statistics/
12. Loupy A, Aubert O, Reese PP, Organ procurement and transplantation during the COVID-19 pandemic: Lancet, 2020; 395(10237); e95-96
13. : Guidance from the International Society of Heart and Lung Transplantation Regarding the SARS CoV-2 Pandemic https://ishlt.org/ishlt/media/documents/SARS-CoV-2_-Guidance-for-Cardiothoracic-Transplant-and-VAD-centers.pdf
14. : ASTS COVID-19 Strike Force Initial Guidance https://asts.org/advocacy/covid-19-resources/asts-covid-19-strike-force/asts-covid-19-strike-force-initial-guidance#.X58VsNBKg2y
15. Picard C, Le Pavec J, Tissot A, Impact of the Covid-19 pandemic and lung transplantation program in France: Respir Med Res, 2020; 78; 100758
16. : Double-Lung Transplant Saves Patient After COVID-19 | Northwestern Medicine https://www.nm.org/healthbeat/medical-advances/covid-19-advances-in-care/double-lung-transplant-saves-patient-after-covid-19
Tables
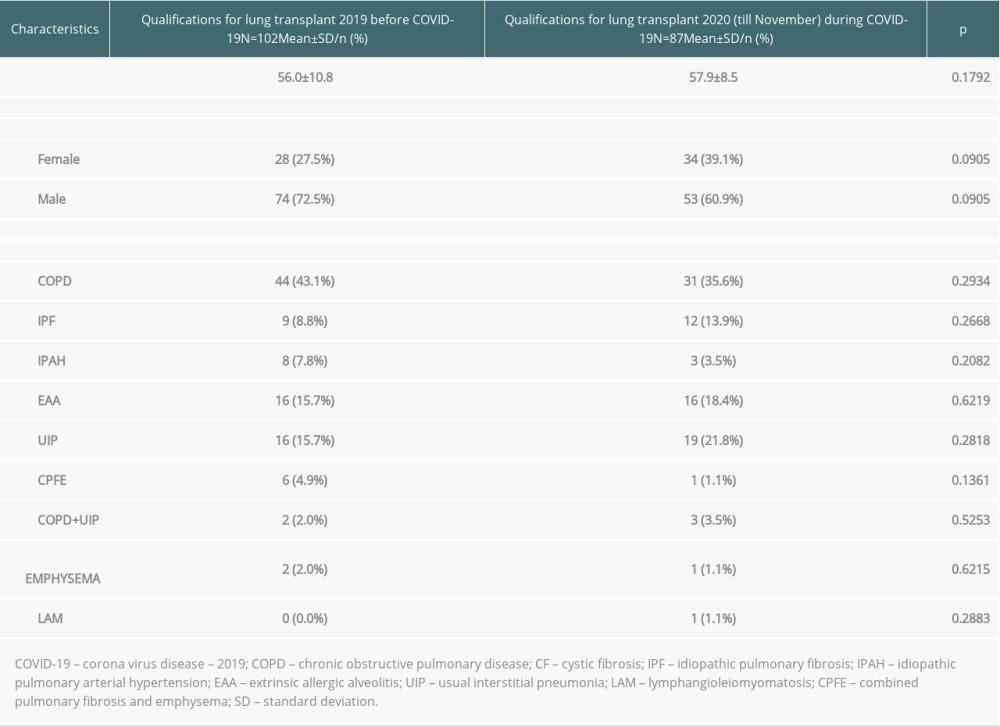 Table 1. Characteristics of patients considered for lung transplantation.
Table 1. Characteristics of patients considered for lung transplantation.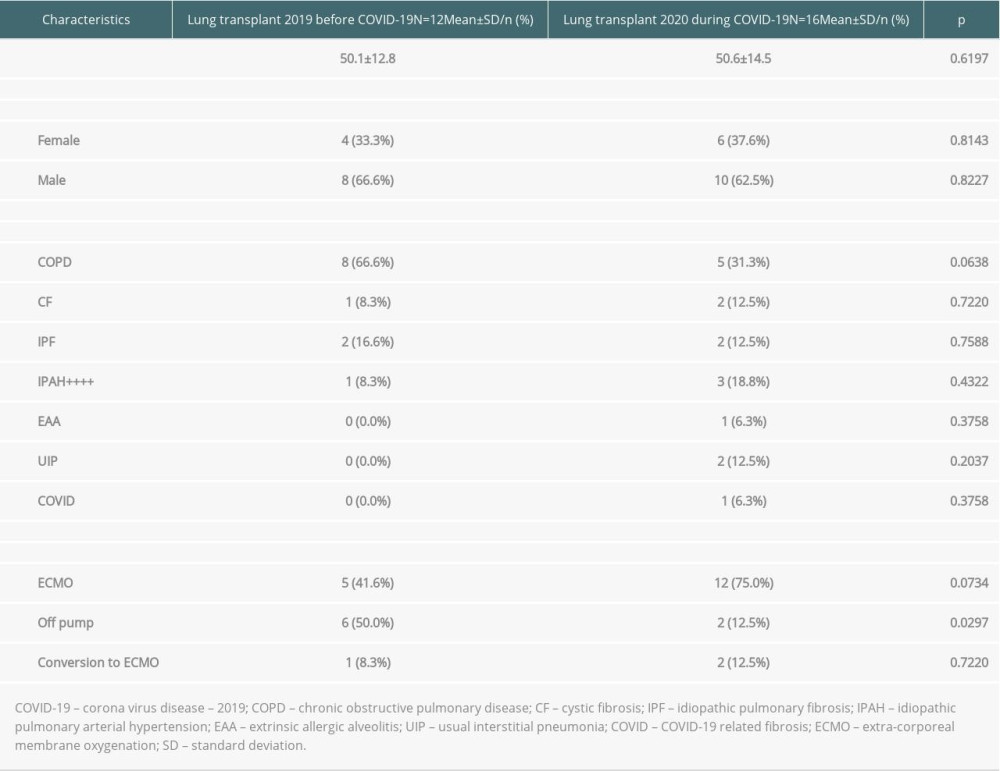 Table 2. Recipient characteristics.
Table 2. Recipient characteristics.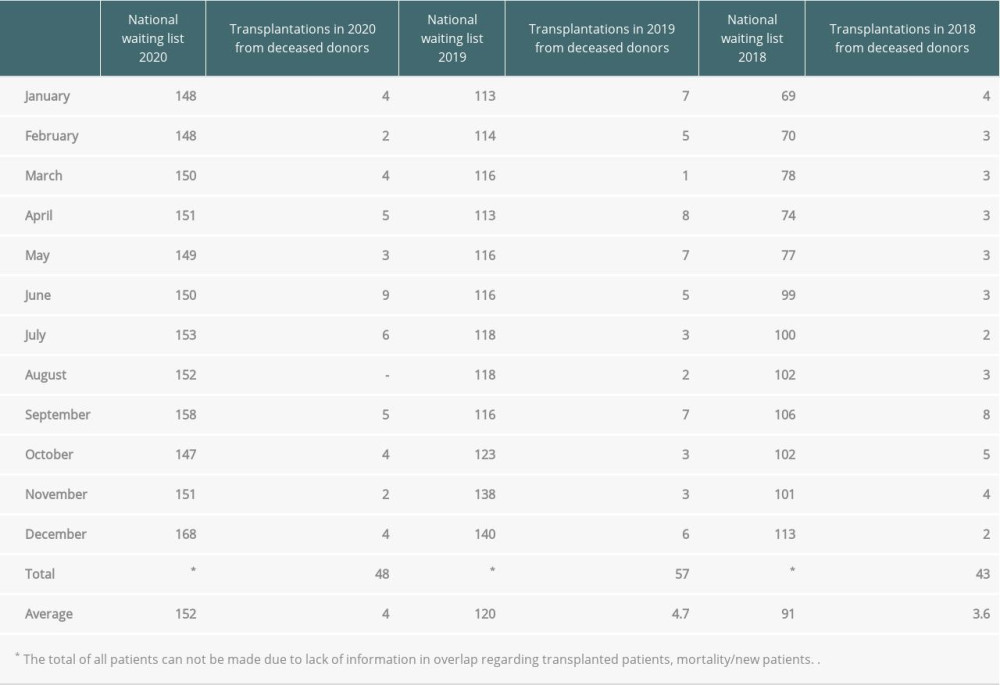 Table 3. Lung transplantation during 2018–2020 in Poland. Data obtained by the Polish Transplant Coordinating Center (Poltransplant) webpage.
Table 3. Lung transplantation during 2018–2020 in Poland. Data obtained by the Polish Transplant Coordinating Center (Poltransplant) webpage. Table 1. Characteristics of patients considered for lung transplantation.
Table 1. Characteristics of patients considered for lung transplantation. Table 2. Recipient characteristics.
Table 2. Recipient characteristics. Table 3. Lung transplantation during 2018–2020 in Poland. Data obtained by the Polish Transplant Coordinating Center (Poltransplant) webpage.
Table 3. Lung transplantation during 2018–2020 in Poland. Data obtained by the Polish Transplant Coordinating Center (Poltransplant) webpage. In Press
15 Mar 2024 : Review article
Approaches and Challenges in the Current Management of Cytomegalovirus in Transplant Recipients: Highlighti...Ann Transplant In Press; DOI: 10.12659/AOT.941185
18 Mar 2024 : Original article
Does Antibiotic Use Increase the Risk of Post-Transplantation Diabetes Mellitus? A Retrospective Study of R...Ann Transplant In Press; DOI: 10.12659/AOT.943282
20 Mar 2024 : Original article
Transplant Nephrectomy: A Comparative Study of Timing and Techniques in a Single InstitutionAnn Transplant In Press; DOI: 10.12659/AOT.942252
28 Mar 2024 : Original article
Association Between FEV₁ Decline Rate and Mortality in Long-Term Follow-Up of a 21-Patient Pilot Clinical T...Ann Transplant In Press; DOI: 10.12659/AOT.942823
Most Viewed Current Articles
05 Apr 2022 : Original article
Impact of Statins on Hepatocellular Carcinoma Recurrence After Living-Donor Liver TransplantationDOI :10.12659/AOT.935604
Ann Transplant 2022; 27:e935604
12 Jan 2022 : Original article
Risk Factors for Developing BK Virus-Associated Nephropathy: A Single-Center Retrospective Cohort Study of ...DOI :10.12659/AOT.934738
Ann Transplant 2022; 27:e934738
22 Nov 2022 : Original article
Long-Term Effects of Everolimus-Facilitated Tacrolimus Reduction in Living-Donor Liver Transplant Recipient...DOI :10.12659/AOT.937988
Ann Transplant 2022; 27:e937988
15 Mar 2022 : Case report
Combined Liver, Pancreas-Duodenum, and Kidney Transplantation for Patients with Hepatitis B Cirrhosis, Urem...DOI :10.12659/AOT.935860
Ann Transplant 2022; 27:e935860