15 December 2021: Case Report
Rex Shunt for Portal Vein Thrombosis After Pediatric Living Donor Liver Transplantation
Yuji Soejima1ABDE*, Tomoaki Taguchi2DE, Toshiharu Matsuura2D, Makoto Hayashida2D, Toru Ikegami1BD, Tomoharu Yoshizumi1BD, Yoshihiko Maehara1DDOI: 10.12659/AOT.909493
Ann Transplant 2021; 26:e909493
Abstract
BACKGROUND: Portal vein thrombosis (PVT) after pediatric liver transplantation (LT) is a common but grave complication which could eventually result in life-threatening portal hypertension. A “Rex” shunt between the superior mesenteric vein and the Rex recess of the liver has been reported to be a treatment option for extrahepatic portal vein obstruction; however, its application to living donor liver transplantation (LDLT) is limited due to the availability of appropriate vein grafts. In this study, we retrospectively evaluated the effectiveness of Rex shunt as an option for the treatment of PVT after pediatric LDLT.
CASE REPORT: Three children underwent the Rex shunt for early (n=2) and late (n=1) PVT after LDLT using the greater saphenous vein (n=2) and the external iliac vein (n=1) from the parents who previously donated their livers. Two of the 3 children are free from symptoms with patent shunt grafts at 14 years after the procedures. One child died at 30 days after LDLT due to repeated episodes of PVT, which finally led to hepatic infarction.
CONCLUSIONS: The Rex shunt is feasible to treat PVT after LDLT. However, additional surgical insults to the living donor need further discussion.
Keywords: Liver Transplantation, Living Donors, Portal Vein, Thrombosis, Child, Humans, Venous Thrombosis
Background
Portal vein thrombosis (PVT) after liver transplantation (LT) is a common problem, especially for pediatric patients such as patients with biliary atresia (BA) with hypoplastic PV. The appropriate treatment for PVT is a very difficult and controversial issue. Some investigators [1,2] believe that recanalization of the PV is not necessary, especially for late PVT without symptoms, provided that hepatopetal collateral vessels develop and liver function is normal. However, recent data [3–5] have shown that late PVT develops in as many as 8.7% of pediatric patients after LT, and it often leads to life-threatening portal hypertension and gastrointestinal bleeding, or even to graft loss if left untreated. Therefore, it has been recognized that a mode of treatment is required for patients with late PVT irrespective of symptoms, especially in pediatric patients [6].
Various treatment options, including surgical and radiological approaches, have been attempted to cure this complication but the results have been suboptimal. However, the recent introduction of the mesenterico-portal bypass or “Rex shunt” [7] causes a paradigm shift in the therapeutic approach to the management of extrahepatic portal vein obstruction (EHPVO) including PVT after LT. However, its application to pediatric living donor LT (LDLT) is limited because it is extremely hard to obtain appropriate vein grafts from small pediatric patients or from deceased donors, especially in countries where the number of diseased donors are limited. Nonetheless, there have been no reports regarding the Rex shunt after LDLT.
We herein report on 3 pediatric patients who underwent the Rex shunt or its variant procedure after LDLT, using homologous vein grafts procured from living donors. Technical details as well as ethical issues are discussed.
Case Report
CASE 1:
A 16-month-old female with post Kasai BA underwent LDLT using a left lateral segment graft donated from her 45-year-old father. The graft weight was 260 g, corresponding to the graft-to-recipient weight ratio of 3.01%. There were no significant collateral veins recognized on preoperative contrast-enhanced computed tomography (CT) scan. The diameter of the recipient PV was 4 mm, for which end-to-end direct anastomosis between the recipient PV trunk and the left PV of the graft using 7-0 PDS continuous sutures for the posterior wall and interrupted sutures for the anterior wall were performed. Appropriate PV flow was confirmed using color Doppler ultrasound intraoperatively. No significant collateral vein was recognized during the procedure. The Doppler signal for the PV became sluggish with a to-and-fro wave form since postoperative day (POD) 7; however, the patient’s general condition and liver function tests were consistently stable. In addition, there was no significant collateral vein which could steal PV flow was recognized on CT scan. Therefore, we decided to follow the patient without relaparotomy. A liver function test showed slightly elevated aspirate aminotransferase (56 U/L) and alanine aminotransferase (34 IU/L) levels on POD 30, for which liver biopsy was indicated. A liver biopsy revealed severe macrovesicular steatosis (>80%), which was attributed to PVT confirmed on angiography. Reestablishment of PV flow was considered to be essential but thrombectomy of the PV was a very difficult option. The umbilical portion (UP) of the PV was confirmed patent by ultrasound. Therefore, a mesenterico-left PV bypass (Rex shunt) using the donor’s left greater saphenous vein (GSV) grafts was planned and performed on POD 49 after LDLT. The infra-pancreatic superior mesenteric vein was exposed and controlled with tape, and all of the tributary veins were ligated. The Rex recess of the left lobe graft was then approached with the round ligament being stretched ventrally. The connective tissue sheath on the UP was dissected and controlled with special care not to injure any PV branches derived from the UP, such as the P3 and P2. The left GSV, which was 10 cm long and 7 mm in diameter, was procured from the same donor. The distal end of the GSV was anastomosed to the recipient superior mesenteric vein using 7-0 PDS continuous sutures and the proximal end was anastomosed end-to-side to the side-clamped UP (Figure 1). PV flow was significantly increased and remained patent thereafter. Liver biopsy 44 days after the shunt procedure showed a marked improvement in steatosis [8]. The patient is well, and the Rex shunt continues to be open at 14 years after the procedure.
CASE 2:
An 11-month-old male with post Kasai BA underwent LDLT using a left lobe graft without the middle hepatic vein donated from his 20-year-old mother. The graft weight was 250 g and the graft-to-recipient weight ratio was 3.33%. There were no significant collateral veins recognized on preoperative contrast-enhanced CT scan. The diameter of the recipient PV was approximately 4.5 mm, for which a patch portoplasty technique using the donor’s inferior mesenteric vein was performed. We performed end-to-end anastomosis between the PV with a patch plasty and the left PV graft using 7-0 PDS continuous sutures for the posterior wall and interrupted sutures for the anterior wall. PV flow was confirmed to be excellent using color Doppler ultrasound intraoperatively. No significant collateral vein was recognized during the procedure. The Doppler signal for PV became to-and-fro from POD 1; however, the signal became hepatopetal thereafter. The patient developed a stomach perforation, for which emergent relaparotomy and simple closure of the perforation were performed. Two days after relaparotomy, liver function tests were markedly elevated and the Doppler signal for the PV disappeared. Angiography revealed PVT, for which a variant of the Rex shunt using the donor’s GSV graft between the recipient left renal vein and the graft UP was created on POD 9, and the patient was on heparin after this time (Figure 2A). In this case, the left renal vein was chosen as an inflow because the superior mesenteric vein (SMV) was also hypoplastic. However, the graft was thrombosed again 5 days after the procedure. We decided to arterialize the PV because it was difficult to maintain adequate PV flow. The splenic artery was dissected distally to the splenic hilum and splenectomy was performed. The splenic artery was flipped over and anastomosed end-to-end to the previous GSV graft with interrupted 8-0 Prolene sutures under a microscope (Figure 2B). Pulsatile PV flow was confirmed immediately after the procedure. However, liver function tests continued to worsen afterwards, and the patient died on POD 30.
CASE 3:
A 1-year-old female with post Kasai BA underwent LDLT using a left lateral segment graft donated from her 28-year-old mother. The graft weight was 260 g and the graft-to-recipient weight ratio was 4.24%. There were no significant collateral veins recognized on preoperative CT scan. The intima of the PV of the recipient was relatively thick but the PV flow from the stump was excellent, for which end-to-end direct anastomosis between the recipient PV trunk and the left PV of the graft was amenable. No significant collateral vein was recognized during the transplantation procedures. Bile leakage from the cut surface developed on POD 3, which required relaparotomy. Acute rejection developed on POD 11, for which steroid pulse therapy was effective. The PV flow was confirmed to be excellent during the postoperative course.
The patient had been very well until 4.5 years after LDLT when she abruptly developed gastrointestinal bleeding. A CT scan revealed thrombosed PV and marked cavernous transformation around the hepatic hilum (Figure 3A). There was no definite bleeding point detected by upper and lower endoscopic examinations. Therefore, the gastrointestinal bleeding was attributed to the ruptured cavernous transformation, for which coiling of the collateral veins was successfully performed through one of the jejunal veins. The patient had been well 3 months after the procedure when gastrointestinal bleeding recurred. At that time, we decided to perform the Rex shunt using the recipient collateral vein. The recipient’s large collateral vein was dissected and flipped over via the retropyloric route and anastomosed end-to-side to the UP with 7-0PDS continuous sutures (Figure 3A). Immediate PV flow was excellent; however, the shunt was thrombosed 3 days after the procedure, for which the emergent Rex shunt using the donor’s left external iliac vein was made between the superior mesenteric vein and the Rex recess (Figure 3B). The patient was on warfarin for 1 year after the procedure. The patient is well without any symptoms and the Rex shunt continues to be open at 14 years after the procedure (Figure 3C and 3D: 3.3 years after the procedure).
In all cases, donors were fully informed of the risks and benefits for the second operation that included taking their GSV (n=2) or the iliac vein (n=1) as vein grafts. Actually, all donors were willing to undergo the second donation, therefore, informed consent was obtained before the procedures.
In total, 3 recipients underwent the procedure for early (n=2, Case 1 and Case 2) and late (n=1, Case 3) PVT after LDLT. The procedure itself was feasible in all 3 cases, however, Case 2 was lost due to repeated episodes of PVT, which finally led to irreversible graft infarction. The other 2 patients are free from symptoms with patent shunt grafts at 14 years after the procedures. No donors experienced complication neither after hepatectomy nor after the second operation.
Discussion
PVT after LT is a common complication, especially after pediatric LT for BA. In a series of LT for BA, the incidence of PV complications was 14.7% [9]. Furthermore, previous studies have revealed that PVT is more commonly seen in partial rather than in whole-size liver grafts [4]. The treatment of PVT, especially for late PVT, is a complicated task. Thrombectomy is the ideal option but it is not usually amenable. Moreover, revision of thrombosed PV is extremely difficult because of the developed collateral cavernous transformation around the hilum. Other options including balloon angioplasty [10,11], portocaval shunting [12], splenectomy [13], and retransplantation [14] have been reported. Some investigators have reported the usefulness of metallic vascular stent placement using interventional angioplasty. However, this is only possible when the guide wire passes the stenotic or thrombosed region. The success rate was reported to be 68.8% out of 16 cases and it was suggested that PVT of more than 1 year with complete obstruction with cavernous transformation was associated with technical failure. Nonetheless, the long-term patency of the stent remains unknown.
The “Rex shunt” was first reported in 1992 by de Ville de Goyet [7] for the treatment of PVT after partial LT. The effectiveness of this procedure for EHPVO has been confirmed by various centers [15–17]. The major advantages of the Rex shunt procedure are the restoration of normal and physiological hepatopetal blood flow, a lack of encephalopathy, and avoidance of secondary pulmonary hypertension. Superina et al [18] recently reported on 34 children who received a Rex shunt, with an overall shunt-patency of 91%, with a follow-up of 1–7 years. Furthermore, a panel of experts regarding the surgical management of EHPVO have recommended the Rex shunt as the first-line treatment for symptomatic EHPVO [4]. In addition, this procedure has been sporadically reported to be effective in the setting of PVT after LT [19,20]. Krebs-Schmitt et al [21] reported on 14 children who underwent the Rex shunt after LT using autologous internal jugular (n=10) or cryopreserved iliac (n=4) vein grafts. They concluded the best result could be obtained with the use of autologous jugular veins.
Regarding to the type of vascular grafts for the Rex shunt, we used the donor’s GSV for small pediatric patients such as our Case 1 and Case 2, and the external iliac vein for relatively large patients such as our Case 3 (6 years of age). In LDLT for small children, the partial liver graft (the left lateral segment graft) usually rotates to the right after implantation and the UP usually locate to the far right. Therefore, a longer vein grafts was necessary for creating a Rex shunt in LDLT. The patients presented here were very small which precluded the use of their own vein grafts. The adult GSV is a strong vein graft and can be potentially longer and more accessible than the external iliac vein. The diameter of the GSV may be sufficient for small pediatric patients. However, the GSV has multiple valves, which mandate the direction of the graft and could potentially be a source of thrombosis formation. We currently believe that the external iliac vein is the most ideal vein graft that is available for PV reconstruction in terms of diameter and length, as well as thickness of the vascular wall. However, the length of the external iliac vein is relatively short (approximately 8 cm), which may preclude its use in larger patients. Furthermore, it requires more invasive procedures and may cause transient postoperative leg edema for otherwise healthy donors. Other options include the internal jugular vein of the recipients, but this is not possible when the recipients are too small with a short neck.
To the best of our knowledge, this report is the first report on the Rex shunt using vein grafts procured from living donors who had previously donated their partial liver. In countries where vascular grafts from deceased donors are rarely available, procurement from the previous donor currently seems to be the best solution. The use of cryopreserved veins is an option, knowing that the quality of the graft is suboptimal for reconstruction of PV.
Ethical issues that expose the living related donors to a second insult should be thoroughly discussed. In this case series, we did not inform any of the donors of the potential second donor operation before LDLT, however, we believe that the procedure is justified as long as the donors show spontaneous consent for the second operation to donate their vascular grafts. However, we currently think it is appropriate to discuss and inform donors of the possibility of second operation before LDLT, especially when the risk of PVT is high. Moreover, it is important to remember that the procedure cannot be performed immediately after the previous hepatectomy because the risk to the donor should be minimal.
Conclusions
The Rex shunt is a valuable surgical option to treat PVT after LDLT when other options are not feasible. However, a longer follow-up is warranted not only for the recipient, but also for the donor.
Figures
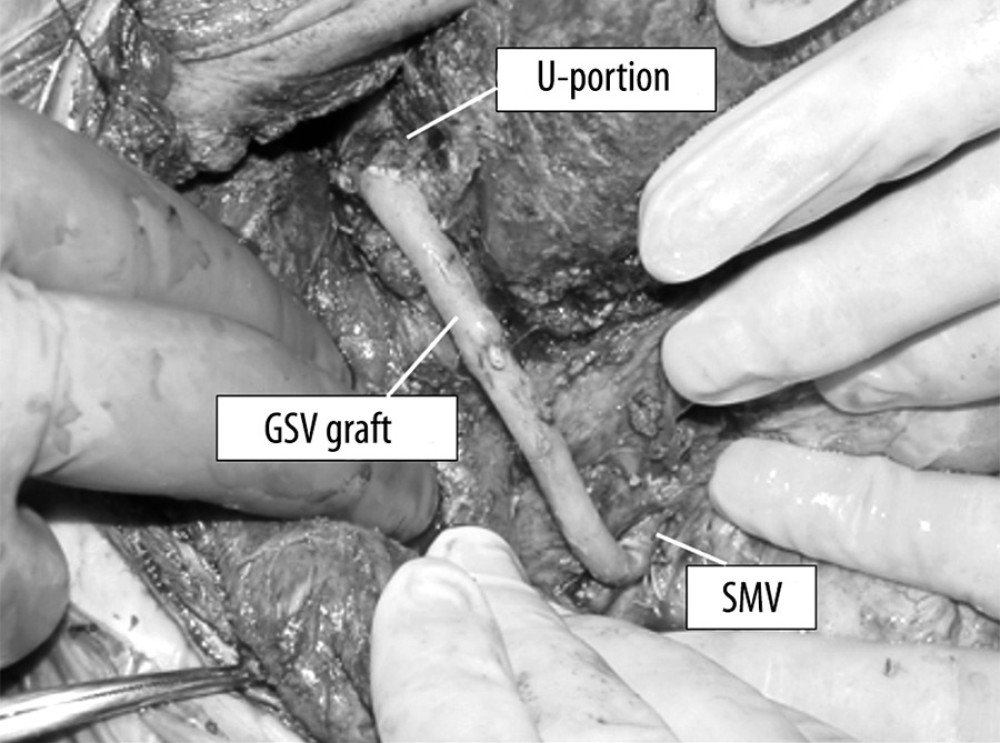 Figure 1. Intraoperative view of Case 1 after creation of the Rex shunt. GSV – greater saphenous vein; SMV – superior mesenteric vein.
Figure 1. Intraoperative view of Case 1 after creation of the Rex shunt. GSV – greater saphenous vein; SMV – superior mesenteric vein. 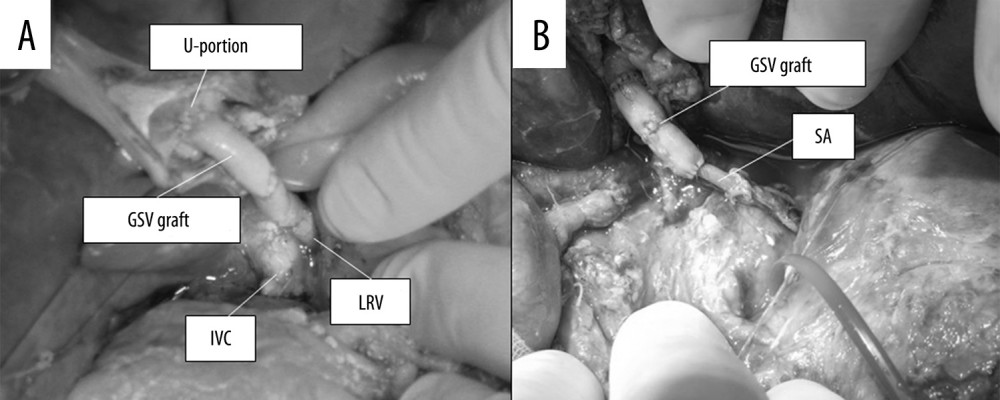 Figure 2. Intraoperative view of Case 2 after creation of the Rex shunt between the LRV and the U-portion of the graft using the donor’s GSV (A) and the SA (B). LRV – left renal vein; GSV – greater saphenous vein; SA – splenic artery; IVC – inferior vena cava.
Figure 2. Intraoperative view of Case 2 after creation of the Rex shunt between the LRV and the U-portion of the graft using the donor’s GSV (A) and the SA (B). LRV – left renal vein; GSV – greater saphenous vein; SA – splenic artery; IVC – inferior vena cava. 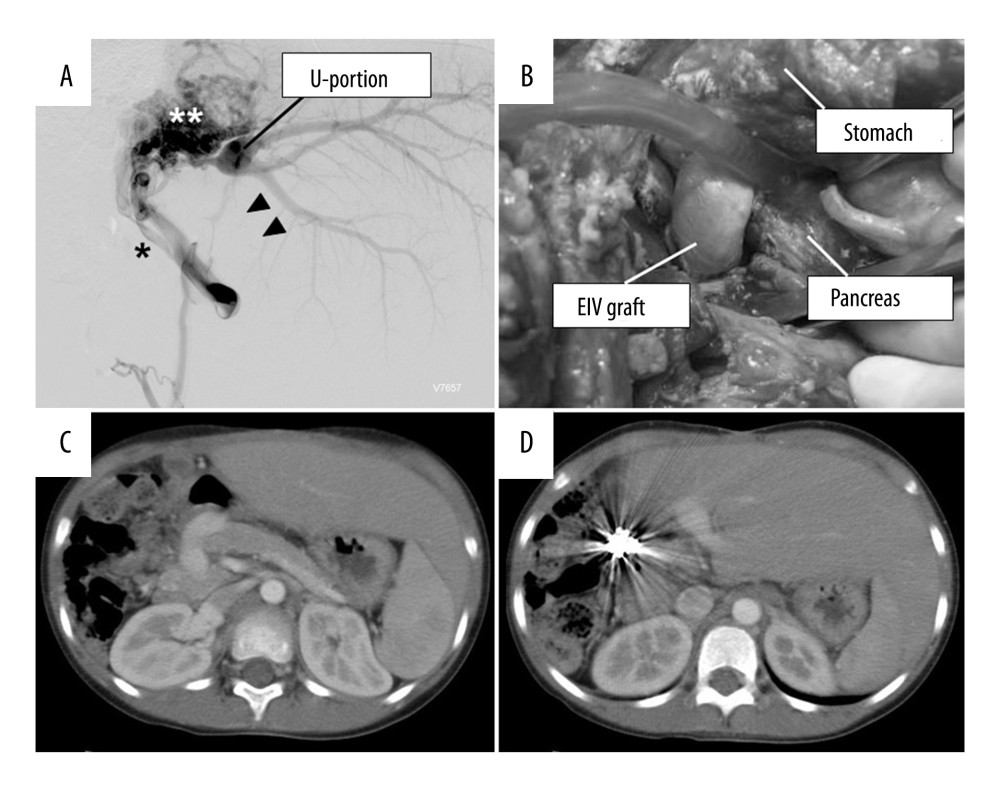 Figure 3. (A) Angiographic findings of portal vein thrombosis of Case 3. Note that the portal vein trunk is completely thrombosed (arrowheads) and a large collateral vein (*) with marked cavernous transformation (**) has formed. (B) Intraoperative view of the Rex shunt created between the SMV and the UP using the donor’s external iliac vein. Note the shunt graft is tunneled through the prepancreatic retropyloric route. (C, D) A computed tomography scan taken at 3.3 years after the procedure shows a patent Rex shunt graft (C) and the UP of the graft (D). EIV – external iliac vein; UP – umbilical portion of the portal vein.
Figure 3. (A) Angiographic findings of portal vein thrombosis of Case 3. Note that the portal vein trunk is completely thrombosed (arrowheads) and a large collateral vein (*) with marked cavernous transformation (**) has formed. (B) Intraoperative view of the Rex shunt created between the SMV and the UP using the donor’s external iliac vein. Note the shunt graft is tunneled through the prepancreatic retropyloric route. (C, D) A computed tomography scan taken at 3.3 years after the procedure shows a patent Rex shunt graft (C) and the UP of the graft (D). EIV – external iliac vein; UP – umbilical portion of the portal vein. References
1. Usuda M, Fujimori K, Koyamada N, Serious intestinal bleeding from vascular ectasia secondary to portal thrombosis after living donor liver transplantation in a child: J Hepatobiliary Pancreat Surg, 2005; 12; 317-20
2. Vannevell G, Clapuyt P, Reding R, Menten R, Spontaneous meso-portal shunt following orthotopic liver transplantation in a child: Pediatr Radiol, 2010; 40(Suppl); S92-94
3. Ueda M, Oike F, Kasahara M, Portal vein complications in pediatric living donor liver transplantation using left-side graft: Am J Transpl, 2008; 8; 2097-105
4. Neto JS, Fonseca EA, Feier FH, Analysis of factors associated with portal vein thrombosis in pediatric living donor liver transplant recipients: Liver Transpl, 2014; 20; 1157-67
5. Cho YP, Kim KM, Ha TY, Management of late-onset portal vein complications in pediatric living-donor liver transplantation: Pediatr Transplant, 2014; 18; 64-71
6. Superina R, Shneider B, Emre S, Surgical guidelines for the management of extra-hepatic portal vein obstruction: Pediatr Transplant, 2006; 10; 908-13
7. De Ville de Goyet J, Clapuyt P, Otte JB, Extrahilar mesenterico-left portal shunt to relieve extrahepatic portal vein obstruction: Transplantation, 1992; 53; 231-32
8. Ogita K, Taguchi T, Soejima Y, Fatty liver caused by portal vein thrombosis after living donor liver transplantation: A case report: J Pediatr Surg, 2005; 40; E7-9
9. Ou HY, Concejero AM, Huang TL, Portal vein thrombosis in biliary atresia patients after living donor liver transplantation: Surgery, 2011; 149; 40-47
10. Yang J, Xu MQ, Yan LN, Management of venous stenosis in living donor liver transplant recipients: World J Gastroenterol, 2009; 15; 4969-73
11. Chen YF, Ou HY, Tsang LLC, Vascular stents in the management of portal venous complications in living donor liver transplantation: Am J Transpl, 2010; 10; 1276-83
12. Emre S, Dugan C, Frankenberg T, Surgical portosystemic shunts and Rex bypass in children: A single-center experience: HPB, 2009; 11; 252-57
13. Tokodai K, Kawagishi N, Miyagi S, Splenectomy for severe intestinal bleeding caused by portal hypertensive enteropathy after pediatric living-donor liver transplantation: A report of three cases: Transplant Proc, 2017; 49; 1129-32
14. de Ville de Goyet J, Alberti D, Falchetti D, Treatment of extrahepatic portal hypertension in children by mesenteric-to-left portal vein bypass: A new physiological procedure: Eur J Surg, 1999; 165; 777-81
15. Fuchs J, Warmann S, Kardorff R, Mesenterico-left portal vein bypass in children with congenital extrahepatic portal vein thrombosis: A unique curative approach: J Pediatr Gastroenterol Nutr, 2003; 36; 213-16
16. Bambini DA, Suprina R, Almond PS, Experience with the Rex shunt (mesenterico-left portal bypass) in children with extrahepatic portal hypertension: J Pediatr Surg, 2000; 35; 13-18
17. Superina R, Bambini DA, Lokar J, Correction of extrahepatic portal vein thrombosis by the mesenteric to left portal vein bypass: Ann Surg, 2006; 243; 515-21
18. de Ville de Goyet J, Gibbs P, Clapuyt P, Original extrahilar approach for hepatic portal revascularization and relief of extrahepatic portal hypertension related to later portal vein thrombosis after pediatric liver transplantation. Long term results: Transplantation, 1996; 62; 71-75
19. Takahashi Y, Nishimoto Y, Matsuura T, Surgical complications after living donor liver transplantation in patients with biliary atresia: Relatively high incidence of portal vein complications: Pediatr Surg Int, 2009; 25; 745-51
20. Gibelli NEM, Tannuri U, de Pinho-Apezzato ML, “Rex shunt” for the treatment of portal vein thrombosis after pediatric liver transplantation: A case report: Transplant Proc, 2009; 41; 955-56
21. Krebs-Schmitt D, Briem Richter A, Grabhorn E, Effectiveness of Rex shunt in children with portal hypertension following liver transplantation or with primary portal hypertension: Pediatr Transplant, 2009; 13; 540-44
Figures
 Figure 1. Intraoperative view of Case 1 after creation of the Rex shunt. GSV – greater saphenous vein; SMV – superior mesenteric vein.
Figure 1. Intraoperative view of Case 1 after creation of the Rex shunt. GSV – greater saphenous vein; SMV – superior mesenteric vein. Figure 2. Intraoperative view of Case 2 after creation of the Rex shunt between the LRV and the U-portion of the graft using the donor’s GSV (A) and the SA (B). LRV – left renal vein; GSV – greater saphenous vein; SA – splenic artery; IVC – inferior vena cava.
Figure 2. Intraoperative view of Case 2 after creation of the Rex shunt between the LRV and the U-portion of the graft using the donor’s GSV (A) and the SA (B). LRV – left renal vein; GSV – greater saphenous vein; SA – splenic artery; IVC – inferior vena cava. Figure 3. (A) Angiographic findings of portal vein thrombosis of Case 3. Note that the portal vein trunk is completely thrombosed (arrowheads) and a large collateral vein (*) with marked cavernous transformation (**) has formed. (B) Intraoperative view of the Rex shunt created between the SMV and the UP using the donor’s external iliac vein. Note the shunt graft is tunneled through the prepancreatic retropyloric route. (C, D) A computed tomography scan taken at 3.3 years after the procedure shows a patent Rex shunt graft (C) and the UP of the graft (D). EIV – external iliac vein; UP – umbilical portion of the portal vein.
Figure 3. (A) Angiographic findings of portal vein thrombosis of Case 3. Note that the portal vein trunk is completely thrombosed (arrowheads) and a large collateral vein (*) with marked cavernous transformation (**) has formed. (B) Intraoperative view of the Rex shunt created between the SMV and the UP using the donor’s external iliac vein. Note the shunt graft is tunneled through the prepancreatic retropyloric route. (C, D) A computed tomography scan taken at 3.3 years after the procedure shows a patent Rex shunt graft (C) and the UP of the graft (D). EIV – external iliac vein; UP – umbilical portion of the portal vein. In Press
18 Mar 2024 : Original article
Does Antibiotic Use Increase the Risk of Post-Transplantation Diabetes Mellitus? A Retrospective Study of R...Ann Transplant In Press; DOI: 10.12659/AOT.943282
20 Mar 2024 : Original article
Transplant Nephrectomy: A Comparative Study of Timing and Techniques in a Single InstitutionAnn Transplant In Press; DOI: 10.12659/AOT.942252
28 Mar 2024 : Original article
Association Between FEV₁ Decline Rate and Mortality in Long-Term Follow-Up of a 21-Patient Pilot Clinical T...Ann Transplant In Press; DOI: 10.12659/AOT.942823
02 Apr 2024 : Original article
Liver Transplantation from Brain-Dead Donors with Hepatitis B or C in South Korea: A 2014-2020 Korean Organ...Ann Transplant In Press; DOI: 10.12659/AOT.943588
Most Viewed Current Articles
05 Apr 2022 : Original article
Impact of Statins on Hepatocellular Carcinoma Recurrence After Living-Donor Liver TransplantationDOI :10.12659/AOT.935604
Ann Transplant 2022; 27:e935604
12 Jan 2022 : Original article
Risk Factors for Developing BK Virus-Associated Nephropathy: A Single-Center Retrospective Cohort Study of ...DOI :10.12659/AOT.934738
Ann Transplant 2022; 27:e934738
22 Nov 2022 : Original article
Long-Term Effects of Everolimus-Facilitated Tacrolimus Reduction in Living-Donor Liver Transplant Recipient...DOI :10.12659/AOT.937988
Ann Transplant 2022; 27:e937988
15 Mar 2022 : Case report
Combined Liver, Pancreas-Duodenum, and Kidney Transplantation for Patients with Hepatitis B Cirrhosis, Urem...DOI :10.12659/AOT.935860
Ann Transplant 2022; 27:e935860








