02 July 2021: Review Paper
Clinical Rationale for a Routine Testing Schedule Using Donor-Derived Cell-Free DNA After Kidney Transplantation
Akshta Pai 1ADEF* , Joshua T. Swan 23ADEF , David Wojciechowski 4ADEF , Yasir Qazi 5ADEF , Sham Dholakia 6ADEF , Grigoriy Shekhtman 6ADEF , Anas Abou-Ismail 6ADEF , Dhiren Kumar 7ADEFDOI: 10.12659/AOT.932249
Ann Transplant 2021; 26:e932249
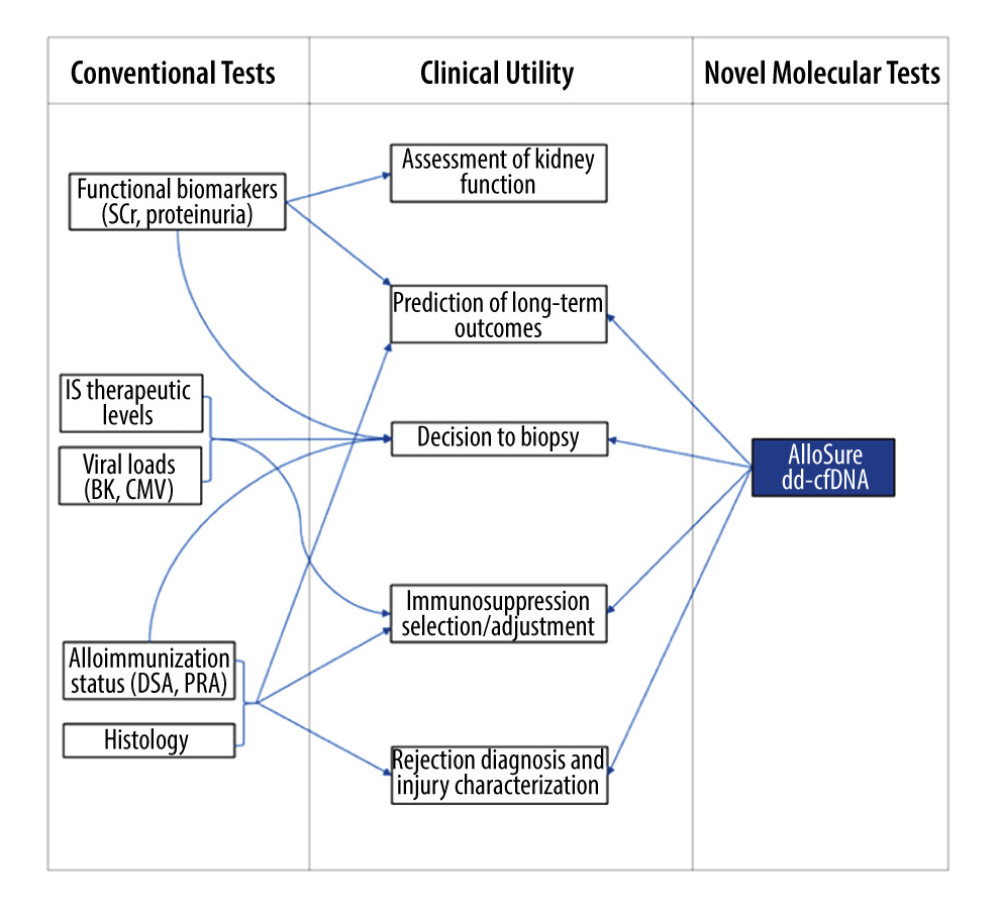
Figure 1 Graphic depicting the potential added value of donor-derived cell-free DNA (dd-cfDNA) surveillance for augmenting clinical decision making in posttransplant management. SCr – serum creatinine; DSA – donor-specific antibodies; PRA – panel reactive antibodies; IS – immunosuppressive therapy.


